| Livestock Research for Rural Development 29 (12) 2017 | Guide for preparation of papers | LRRD Newsletter | Citation of this paper |
Endoparasitism is among important causes of reduced productivity of indigenous chickens. This study was undertaken to determine the prevalence and intensity of gastrointestinal (GI) parasites in apparently healthy free range chickens slaughtered at various live bird markets in Nairobi County, Kenya. The 131 sampled chickens of both sexes originated from 8 counties with varying agro-climatic conditions. Worm egg and coccidial oocyst counts were performed on fecal materials from each bird while worms collected from the GI tracts were identified and quantified. Many chickens had adult GI helminths (90%), but only a few of these (14%) had coccidial oocysts. Nematodes were the predominant helminths (89%) followed by cestodes (51.5%) but no trematodes were recovered. The nematodes recovered were: Heterakis isolonche (59.5%), Subulura brumpti (36%), Tetrameres americana (32%),Ascaridia galli (19.8%), Gongylonema ingluvicola (19.1%),Acuaria hamulosa (6.1%), Heterakis gallinarum (5.3%) andCapillaria spp. (2.3%). The cestodes recovered were: Raillietina echinobothrida (37.7%), Hymenolepis carioca (33.6%), Davainea proglottina (6.9%), R. tetragona (6.1%) and R. cesticillus (2.3%). Other endoparasites encountered were the air-sac mite, Cytodites nudus (15.3%), Sacocystis spp. (5.6%) and Syngamus trachea (4%). The results of this study showed that GI parasites are prevalent and may pose health problems in apparently healthy free range chickens. The parasites economic importance should be investigated and control strategies initiated to improve the health and productivity of indigenous chickens in Kenya.
Key words: Ascaridia galli, counties, helminths, Heterakis spp., Raillietina spp. sexesIndigenous chickens are defined as a group of heterogeneous native fowls, which have been left to out-cross over the years in rural households (Tuitoek et al 1999) and their importance for rural economy is immense in many countries of the world (Barua and Yoshimura 1997; Vali 2008; Magothe et al 2012). These chickens play a major role for the rural poor with respect to their subsidiary income and also provide them with high protein food (eggs and meat). Their potential is enormous and has not been realized because of many constraints like infectious diseases, parasitism, predation, lack of feed, housing, low genetic potential, lack of marketing policy and inadequate farmer education (Nzioka 2002). Parasitism due to GI helminths and ectoparasites constitutes among the major causes that decrease productivity of chickens, but neglected as they are rarely lethal (Permin et al 1997; 2002; Hunduma et al 2010).
Studies conducted on rural free-range poultry in Botswana (Mushi et al 2000), Ethiopia (Zeryehun and Yohannes 2015), Ghana (Poulsen et al 2000), Morocco (Hassouni and Belghyti 2006), Nigeria (Idika et al 2016), Tanzania (Permin et al 1997; Magwisha et al 2002) and Zimbabwe (Mukaratirwa et al 2001; Permin et al 2002) have shown high prevalence of both external and internal parasites among indigenous chickens. In Kenya, Sabuni et al (2010; 2011; 2013) and Waruiru et al (2017) have shown that ecto- and haemo-parasites are common in free range chickens and ducks. However, few such studies have been done on helminth parasites of poultry in the country (Ondawsy et al. 2000; Irungu et al. 2004; Okech et al 2006; Chege et al 2015).
Scavenging free-range birds are in constant contact with soil which serves as an important reservoir and transmission site for infective larval stages of helminths and arthropods which act as paratenic or intermediate hosts (Pandey and Jiang 1992). These factors explain the presence of wide range of helminths and their impact in the health and growth of scavenging rural chickens (Permin et al 1997; Phiri et al 2007). A study conducted in Kenya by Ndegwa et al (1998) showed that farmers did not deworm their indigenous chickens and were not aware about the existence of parasitism in poultry. This study was designed to investigate the prevalence and intensity of GI parasites in marketed indigenous chickens with the ultimate aim of developing control strategies relevant for free range poultry management.
The chickens were purchased from live bird open-air markets located in various trading centers of Nairobi County, Kenya. These were selected using simple random method and included: Burma/Maziwa (Jogoo road), Kariokor, Nairobi South (B and C) and Westlands trading centers. The free range village chickens are brought in cages from different rural areas of Kenya and are usually sold live. However, some consumers prefer to have the chickens slaughtered at the markets in which facilities are readily available. It was established through a questionnaire administered to the traders that sampled birds came from the former Eastern (Machakos, Makueni and Kitui counties), Rift Valley (Bomet, Kericho and Uasin Gishu counties) and Nyanza (Kisii and Homa bay counties) provinces of Kenya, respectively. Counties in the eastern region are in agro-climatic zones 3 to 6 and with a mean annual rainfall of less than 1100 mm. The other counties are in agro-ecological zones 1 to 3 and with a mean annual rainfall of more than 1700 mm (Sombroek et al 1982).
A total of 75 live chickens, mainly adults and a few growers were purchased while 56 other samples were obtained as parts of GI tracts from slaughter slabs within the trading centers.
The GI tracts of the live chickens were obtained during the post mortem examination and separated into various parts namely: crop, proventriculus, gizzard, duodenum, small intestines (jejunum and ileum), caecum and large intestines. Each part was opened and contents emptied into separate containers. They were washed with water to retrieve all the worms clinging on the mucosa. The nematodes and cestodes from these parts were collected and put in separate containers with 70% ethanol. All the nematodes from each anatomical location were quantified. The helminths were processed according to procedures outlined by Gibbons et al (1996), and identified using helminthological keys of Soulsby (1982), Permin and Hansen (1998) and Norton and Ruff (2003).
A fecal sample was obtained from the cloaca or the contents of the large intestines from each bird during post mortem examination. This sample was used to determine fecal egg and coccidial oocyst counts using a modified McMaster technique (MAFF, 1986).
The data derived from these procedures was entered in Ms excel and exported to Instat® (2004) for statistical analysis. Analysis of variance using non-parametric one-way ANOVA (Conover 1980) for sex and place of origin was performed. The prevalence of GI parasites was defined as described by (Margolis et al 1982) and a critical probability of 0.05 was adopted as a cut-off point for statistical significance.
Of the 131 chickens sampled, 118 had one or more adult species of GI helminths giving an overall prevalence of 90%. These chickens harbored an average of 37 worms per chicken with a range of 0-374 worms. Nine nematodes species were recovered namely: Heterakis isolonche (59.5%), Subulura brumpti (35.9%), Tetrameres americana (32.3%), Ascaridia galli (19.8 %), Gongylonema ingluvicola (19.1%), Acuaria hamulosa (6.1). H. gallinarum (5.3%) and Capillaria spp. (2.3%). Five cestodes species recovered included: Raillietina echinobothrida (37.4%),Hymenolepis carioca (33.6%), Davainea proglottina (6.9%), R. tetragona (6.1%) and R. cesticillus (2.3%) (Table 1). No trematodes were recovered in this study. Other endoparasites observed were Syngamus trachea (4.0%), which was identified in histological sections of lung tissues, the mite, Cytodites nudus (15.3%) in the air sacs and Sarcocystis spp. (5.6%) was identified in the neck muscles during microscopic examination.
|
Table 1. Type of helminth, its location in the body and prevalence (%) in marketed indigenous chickens |
|||
|
Worm species (n=131) |
Location of worms in GI tract |
No. of infested |
Prevalence |
|
Gonevlonema ingluvicola |
Crop |
25 |
19.1 |
|
Tetrameres americana |
Proventriculus |
42 |
32.1 |
|
Acuaria hamulosa |
Gizzard |
8 |
6.1 |
|
Ascaridia galli |
Duodenum and small intestines |
26 |
19.8 |
|
Capillaria spp. |
Caecum |
3 |
2.3 |
|
1 Heterakis gallinarum |
Caecum and large intestines |
7 |
5.3 |
|
1 Heterakis isolonche |
Caecum and large intestines |
78 |
59.5 |
|
1 Subulura brumpti |
Caecum and large intestines |
47 |
35.9 |
|
Davainea proglottina |
Duodenum |
9 |
6.9 |
|
Hvmenolepis carioca |
Small intestines |
44 |
33.6 |
|
Raillietina echinobothrida |
Small and large intestines |
49 |
37.7 |
|
Raillietina tetragona |
Small and large intestines |
8 |
6.1 |
|
Raillietina cesticillus |
Large intestines |
3 |
2.3 |
|
1
Mean caecal worm counts were significantly different
(p < 0.05) between counties; |
|||
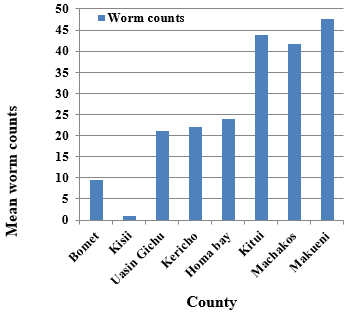
The range of GI nematodes was as follows: T. americana (0-6), A. hamulosa (0-11), A. galli (0-12) and caecal worms ( H. gallinarum, H. isolonche and S. brumpti) 0-374 per chicken. The overall mean intensity of caecal worms was 36.3 and there was a significant difference in mean worm load between counties of bird’s origin (Fig. 1) but not between sexes.
Nineteen percent (25/131) of the fecal samples processed were positive for helminth parasite eggs while 14% (18/131) were positive for coccidial oocysts. The egg types encountered were: Ascaridia spp. (13.3%), Heterakis spp. (13.3%), Capillaria spp. (33.3%) and cestode eggs (40%).
 |
| Figure 1. Mean caecal worm counts in chickens originating from eight different counties of Kenya |
The present study disclosed a high infestation prevalence (90%) of GI parasitism in indigenous chickens in Kenya. These findings are comparable with previous reports (Ondawsy et al 2000; Irungu et al 2004; Okech et al 2006; Chege et al 2015) from different regions of Kenya. The prevalence rate of GI parasites of scavenging chickens was reported to be 90.21% in Ethiopia (Ashenafi and Eshetu 2004) and 100% in Zimbabwe (Jensen and and Pandy 1989). The parasites recorded were mainly helminths and comprised of nematodes and cestodes.
The high prevalence of GI helminth observed in free-range chickens in Kenya may be attributable to the type of production system, as proposed by Magwisha et al (2002). Under this system, local chickens roam from place to place usually seeking for food in the superficial layers of the soil. The soil is often contaminated with living organisms including those that serve as paratenic or intermediate hosts for helminths that infest poultry (Pandey and Jiang 1992; Ashenafi and Eshetu 2004).
Several nematode species as encountered in this study have been reported in variable numbers in indigenous chickens in tropical countries (Permin et al 1997; Poulsen et al 2000; Mukaratirwa et al 2001; Ashenafi and Eshetu 2004; Idika et al 2016) including Kenya (Irungu et al 2004; Chege et al 2015). The most prevalent nematodes were H. isolonche, S. brumpti and T. americana while, A. hamulosa, H. gallinarum and Capillaria spp. were recorded at lower prevalences. This was in contrast to earlier findings of a study conducted in Kakamega County where only four nematode species were reported, with H. gallinarum being the most prevalent followed by A. galli (Ondawsy et al 2000). Five more nematode species (i.e.,H. isolonche, S. brumpti, A. hamulosa. G. ingluvicola and Capillaria spp.) were recorded in the present study of 8 counties in Kenya. Strongyloides avium had been recorded in a previous study in Kenya (Irungu et al. 2004). Heterakis spp. and S. brumpti were isolated from the caecum and large intestines, although their predilection site is the caecum (Norton and Ruff, 2003). This observation was unusual and could be attributed to translocation of the worms from the caecum.
Five tapeworm species were recovered in this study with the most prevalent being R. echinobothrida followed byH. carioca, D. proglottina, R. tetragona and R. cesticillus.This was in agreement with the findings of Chege et al (2015) but differed from those of Ondawsy et al (2000) who encountered only one species, R. tetragona. Other studies conducted in East Africa recorded up to 10 species of tapeworms (Permin et al 1997).
Trematodes were not observed in this study and was in agreement with findings of Chege et al (2015) in Kenya, Yoriyo et al (2008) in Nigeria and Komba et al (2013) in Tanzania. However, other studies conducted in Kenya (Ondawsy et al 2000, Kyalo 2012), Uganda (Ssenyonga 1982) and Bagladesh (Islam et al 2004) recorded the presence of trematodes in chickens.
The study showed that caecal worm loads were significantly higher in warmer counties of Machakos, Makueni and Kitui compared to colder ones of Bomet, Kericho, Kisii and Uasin Gichu. It was also evident that some nematode worms like A. galli. T. americana, S. brumpti, H. isoloche and the cestode, R. echinobothrida were prevalent in all the counties. These variations could be due to differences in local environmental conditions (i.e., temperature, precipitation, humidity etc), which support larval development and facilitate transmission of these helminth parasites (Ashenafi and Eshetu 2004; Kyalo 2012).
The mean intensity of helminth infestations did not differ significantly between male and female chickens as was reported in studies conducted in Nigeria (Yoriyo et al 2008) and Morocco (Hassouni and Belghyti 2006). However, this was in variance with recent findings of Kyalo (2012) in Kenya. Mixed GI helminths and coccidia (Eimeria spp.) infestations were common and this may play a role in early chick mortality and other production losses among adults due to their combined devastating effects on the host metabolism (Nnadi and George 2010).
This study was financially supported by the DANIDA-LHRP funds, Dean’s grant No. 655-466 and ENRECA program on improving the health and productivity of the rural poultry in Africa grant No. 500-661-092.
Ashenafi H and Eshetu 2004 Study on gastrointestinal helminths of local chickens in Central Ethiopia. Revue de Médecine Vétérinaire 10 504-507.
Barua A and Yoshimura Y 1997 Rural poultry keeping in Bangladesh. World’s Poultry Science Journal 53 (4): 392–394.
Chege H W, Kemboi D C, Bebora L C, Maingi N, Mbuthia P G, Nyaga P N, Njagi L W and Githinji J 2015 Studies on seasonal prevalence of ecto- and endo-parasites in indigenous chicken of Mbeere Subcounty, Kenya. Livestock Research for Rural Development. Volume 27, Article #113. Retrieved August 9, 2017, from http://www.lrrd.org/lrrd27/6/cheg27113.htm
Conover W J 1980 Practical Nonparametric Statistics (2nd ed). John Wiley & Sons. pp. 225–226.
Gibbons L M, Jones A and Kahlil L F 1996
Laboratory Manual for Eighth International
Training Course on Identification of Parasites of Economic Importance. CABI
Institute of of Parasitology, UK.
Hassouni T and
Belghyti D 2006
Distribution of gastrointestinal helminths in chicken farms
in the Gharb region, Morocco. Parasitology Research 99 (2): 181-183
Hunduma D, Regassa C, Fufa D, Endale B and Samson L 2010 Major constraints and health management of village poultry production in Rift Valley of Oromia, Ethiopia. American-Eurasian Journal of Agriculture and Environmental Sciences 9: 529-533.
Idika I K, Obi C F, Ezeh I O, Iheagwam C N, Njoku I N and Nwosu C O 2016 Gastrointestinal helminth parasites of local chickens from selected communities in Nsukka region of south eastern Nigeria. Journal of Parasitic Diseases 40 (4) 1376–1380
Instat+ for windows 2004 Version 2.4, Statistical Services Centre, University of Reading, UK.
Irungu, LW, Kimani, RN, Kisia, SM, 2004. Helminth parasites in the intestinal tracts of indigenous poultry in parts of Kenya. Journal of South African Veterinary Association 75: 58-59.
Islam M J, Rahmarv M. S, Talukder M H, Rahrnan M H and Howlider M A R 2004 Investigation into parasitic infestation of scavenging chickens in Bangladesh. The Bangladesh Veterinarian 21 (2): 74-80.
Jensen J and Pandy V S 1989 Observation on helminths parasites of domestic fowls in Zimbabwe. Zimbabwe Veterinary Journal 20 (1):15-17
Komba E V G, Mkupasi E M, Mwesiga G K, Mbyuzi A O, Busagwe Z, Mzula A, Lupindu A M and Nzalawahe J 2013 Occurrence of helminths and coccidia in apparently healthy free range local chickens slaughtered at Morogoro live bird market. Tanzania Veterinary Journal 28 (2) 55-61
Kyalo, M M 2012 Prevalence, intensity and pathology associated with helminth infections in indigenous chickens in Kiambu and Nairobi counties, Kenya. University of Nairobi Archive.
MAFF 1986 Ministry of Agriculture, Fisheries and Food (MAFF). Manual of Veterinary Parasitological Laboratory Techniques, 3rd edition, reference book 418. HMSO, London.
Magothe T M, Okeno T O, Muhuyi W B and Kahi A K 2012 Indigenous chicken production in Kenya: I. Current status. World’s Poultry Science Journal 68 (1): 119–132.
Magwisha H B Kassuku A A Kvysgaard N C and Permin A 2002 A comparison of the prevalence and burdens of helminth infections in growers and adult free-range chickens Tropical Animal Health and Production 34: 205-214.
Margolis L, Esch, G W, Holmer J C, Kuris A M and Schad G A 1982 The use of ecological terms in parasitology. (Report of an ad hoc Committee of the American Society of Parasitologists. Journal of Parasitology 68: 131-133.
Mukaratirwa S, Hove T, Esmann J B, Hoj C J, Permin A and Nansen P 2001 A survey of parasitic nematode infections of chickens in rural Zimbabwe. Onderstepot Journal of Veterinary Research 68:183-186.
Mushi E Z, Binta M G , Chabo R G , Ndebele R and Thibanyane T 2000 Helminth parasites of indigenous chickens in Oodi, Kgatleng District, Botswana. Journal of South African Veterinary Association71(4): 247-248
Ndegwa J M, Kimani C W, Siamba D N, Mukisira E A and Re Jong R 1998 Characteristics of rural poultry production in different agro-ecological zones in Kenya. In: Proceedings of the 6th biennial Kenya Agricultural Research Institute (KARI) scientific conference, 9-13 November 1998, Nairobi, Kenya. pp. 540-547.
Nnadi P A and George S O 2010 A cross-sectional survey on parasites of chickens in selected villages in the subhumid zones of south-eastern Nigeria. Journal of Parasitology Research 2010: 1-6.
Norton R A and Ruff M D 2003 Nematodes and Acanthocephalans. In: Saif Y M, Barnes H J, Glisson J R, Fadly A M, McDougald L R and Swayne D E (Eds). Diseases of Poultry. 11th edition. Iowa State Press, a Blackwell Publishing Company, Ames, Iowa. pp. 931-961.
Nzioka M 2000 Indigenous poultry production in the Katumani mandate district, constraints and prospects. In the Proceedings of the End of Project Conference, Kenya Agricultural Research Institute, Nairobi, Kenya. pp. 213-226.
Okech B A, Irungu L W and Cooper J E 2006 Helminthiasis in free-range indigenous domestic poultry in Kenya. Bulletin of Animal Health and Production in Africa 54: 92-99.
Ondawsy H O, Okitoi L O, Obali M P, Simwa S M and Wakhusama, S M 2000 Epidemiology of helminthes in indigenous chicken in western Kenya: Proceedings of the 7th scientific conference, collaborative research for sustainably improved livelihoods. 13th -17th Nov. 2000. pp. 407-412.
Padhi M K 2016 Importance of indigenous breeds of chicken for rural economy and their improvements for higher production performance. Scientifica 2016: 1-9.
Permin A, Esmann J B, Hoj C H, Hove T and Mukaratirwa S 2002 Ecto-, endo- and haemoparasites in free range chickens in Goromonzi District, Zimbabwe. Preventive Veterinary Medicine 54: 213-224.
Permin A and Hansen J W 1998 Epidemiology, Diagnosis and Control of Poultry Parasites. FAO, Rome, Italy.
Permin A, Magwisha H, Kassuku A A, Nansan P, Bisgaard M, Frandsen F, Gibbons L, 1997 A cross sectional survey of helminths in rural scavenging poultry in Tanzania in relation to season and climate. Journal of Helminthology 71: 233-240.
Poulsen J, Permin A, Hindsbo O, Yelifari L, Nansen P and Bloch P 2000 Prevalence and distribution of gastro-intestinal helminths and haemoparasites in young scavenging chickens in the upper eastern region of Ghana. Preventive Veterinary Medicine 45: 237-245.
Phiri I K, Phiri A M, Ziela M Chota A Masuku M and Monrad J 2007 Prevalence and distribution of gastrointestinal helminths and their effects on weight gain in free-range chickens in Central Zambia. Tropical Animal Health and Production 39 (4): 309-315.
Sabuni Z A, Mbuthia P G, Maingi N, Nyaga P N, Njagi L W, Bebora L C and Michieka J N 2013 Skin lesions associated with ectoparasitic infestation in indigenous chickens in Eastern Province of Kenya. Research Journal of Poultry Sciences 6: 53-58.
Sabuni Z A, Mbuthia P G, Maingi N, Nyaga P N, Njagi L W, Bebora L C and Michieka J N 2011 Prevalence of haemoparasites infection in indigenous chicken in Eastern Province of Kenya.Livestock Research for Rural Development. Volume 23, Article #238. Retrieved January 9, 2017, from http://www.lrrd.org/lrrd23/11/sabu23238.htm
Sabuni Z A, Mbuthia P G, Maingi N, Nyaga P N, Njagi L W, Bebora L C and Michieka J N 2010 Prevalence of ectoparasites infestation in indigenous free-ranging village chickens in different agro-ecological zones in Kenya. Livestock Research for Rural Development. Volume 22, Article #212. http://www.lrrd.org/lrrd22/11/sabu22212.htm
Sombroek W C, Braun H M H and van der Pour B J A 1982 Explanatory soil map and agro-climatic zone map of Kenya. Report E1. National Agricultural Laboratories, Soil Survey Unit, Nairobi, Kenya. p 56.
Ssenyonga G S 1982 Prevalence of helminth parasites of domestic fowl (Gallus domesticus) in Uganda. Tropical Animal Health and Production 14 (4): 201-204.
Tuitoek J K, Chemjor W, Ndegwa J M and Ottaro J M (1999) Morphological characteristic and protein requirements of Kenyan indigenous chickens. In: Proceedings of the 6th biennial Kenya Agricultural Research Institute (KARI) scientific conference, 9-13 November 1998, Nairobi, Kenya. pp. 1-9.
Vali N 2008 Indigenous chicken production in Iran: a review. Pakistan Journal of Biological Sciences 11 (22): 2525–2531.
Waruiru R M, Mavuti S K, Mbuthia P G, Njagi L W, Mutune M N and Otieno R O 2017 Prevalence of ecto-and haemo-parasites of free-range local ducks in Kenya. Livestock Research for Rural Development. Volume 29, Article #126. Retrieved July 4, 2017, from http://www.lrrd.org/lrrd29/7/stek29126.htm
Yoriyo K P, Adang K, Fabiyi J P and Adamu S U 2008 Helminthes parasites of local chickens in Bauchi State, Nigeria. Science World Journal 3 (2) 35-37.
Zeryehun T and Yonas Yohannes Y (2015) Ectoparasite infestation of free scavenging chickens reared under traditional backyard production system in Wolayita Zone, southern Ethiopia. Ethiopian Veterinary Journal 19 (2): 55-66.
Received 22 August 2017; Accepted 27 August 2017; Published 1 December 2017