 |
|
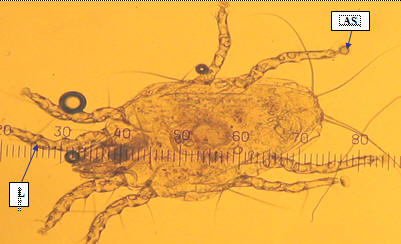
Figure 1. Male Epidemoptes species obtained from a chicken from lower midland 5 showing the first pair of legs (L) which is slightly thicker and ambulatory sucker with short unsegmented stalk (AS) (magnification X40) |
| Livestock Research for Rural Development 22 (11) 2010 | Notes to Authors | LRRD Newsletter | Citation of this paper |
Ectoparasitism is an important factor associated with poor production of village indigenous chickens. A cross-sectional study was carried out to determine the prevalence of ectoparasites in free ranging indigenous chicken from two different agro-ecological zones: Lower highland 1 (LH1) in Embu District and Lower midland 5 (LM5) in Mbeere District, Kenya. A total of 144 chickens of matched age (chicks, growers and adults) and sex groups were examined for the presence of ectoparasites. Of these, 138 (95.8%) had one or more types of ectoparasites, namely; lice, mites, fleas and soft ticks.
One thirty one birds had lice, 107 mites, 42 sticktight fleas and 8 had soft ticks. Of the 138 infested birds, 25 had single while 113 had mixed infestations. Lice were the most prevalent parasites. The study documents Epidermoptes species, Laminosioptes cysticola and Megninia species for the first time in Africa as well as Lipeurus caponis and Goniodes gigas in Kenya. All adult birds were infected with ectoparasites followed by 97.7% grower and 89.6% chicks. Both male and female birds had same prevalence (95.8%) of ectoparasites. Lower midland 5 had a slightly higher prevalence of ectoparasites (98.6%) compared to LH1 (93.1%) though not statistically significant. Parasite intensity was significantly different among age groups of chicken and between agro-ecological zones (p<0.05), but not between sexes of birds (p>0.05).
Because of the high prevalence of ectoparasites revealed by this study, it is imperative that integrated control strategies need to be put in place to improve chicken productivity and enhance smallholder livelihood in these areas.
Key words: Ages, fleas, intensity, lice, mites, sexes, ticks
Indigenous chickens constitute over 81% of poultry in Kenya and produce 71% of eggs and poultry meat (Kiptarus 2005). However, the overriding constraint to expansion and increased productivity of indigenous free-range poultry is their frequent decimation by viral diseases especially Newcastle disease, ecto -, endo - and haemoparasitic infections, predation, theft, other diseases (bacterial and fungal diseases) and low levels of animal health and husbandry practices resulting in high mortality rates especially in chicks (Njunga 2003).
Ectoparasitism negatively affects the productivity potential of the local free-range chickens since they either compete for feed or cause distress to the birds. These parasites are common in rural areas practicing free-range poultry systems since there is inappropriate housing and lack of appreciable pest control efforts (Mungube et al 2006), whereas they are usually controlled in birds in commercial systems. These parasites may constitute a clinical problem; transmit a number of infectious diseases and can also act as transport/ intermediate hosts of a range of helminth parasites (Arends 2003). Moyer et al (2002) noted that a parasite's potential effect, or "pressure", can influence the life history strategy of its host. In environments with high parasite pressure, hosts invest more in anti-parasite defense, which may limit their investment in other life history components, such as survival (Moyer et al 2002) and production.
No previous studies have been carried out in Kenya to determine type, prevalence and intensity of ectoparasitic infestation based on age and sex of the bird; and agro-ecological disposition. The aim of this study was therefore to determine the type, prevalence and intensity of ectoparasites affecting different ages and sex groups of free-range village indigenous chicken from two agro-ecological zones: LH1 in Embu District and LM5 in Mbeere District, Kenya.
Two agro-ecological zones in two neighboring districts were chosen for this study. The selection was based on the availability of free-ranging village indigenous rural poultry population in the areas and contrasting agro-ecological zones.
Of these two, one was a lower highland 1 (LH1) in Embu District. This is a high agricultural potential area where tea, maize, beans and various fruits are grown and free-range poultry and dairy cattle are kept. The area has a bimodal rainfall pattern of long rains between March and June, and short rains in October to December. It has an annual average rainfall of 1080mm. Altitude ranges from 1500 to 4500 meters above sea level. The temperatures range from 12 to 27°C (Onduru et al 2002).
The other study area was the lower midland 5 (LM5) in Mbeere District. This is a semi-arid area with livestock (beef cattle, sheep and goats), poultry, millet and green gram as the main agricultural activities. It has a bimodal and erratic rainfall pattern with average annual rainfall of 180mm per year. Altitude is 1200M above sea level and temperatures range from 20-30°C (Onduru et al 2002).
Indigenous chickens were obtained from individual homesteads using a purposive sampling method. The calculated sample size was 144 birds which were purchased from individual homesteads. The expected prevalence used in the sample size calculation was 50% and the maximum limits of error at 8.3% as per the following formula n=1.962pq/L2 where n is the sample size, p the prevalence, q=1−p and L the limits of error on the prevalence (Martin et al 1987).
The birds were categorized into three age groups, namely: chicks (aged < 2 months), growers (2 to 8 months) and adult (aged > 8 months) according to Magwisha et al (2002) with modification. A total of 72 birds were sampled per agro-ecological zone comprising 36 birds per sex group and 24 birds per age group. All the birds from the two agro-ecological zones were purchased from January to February 2007 in a one month period. They were transported alive in cages to the Department of Veterinary Pathology, Microbiology and Parasitology laboratories, Kabete for examination.
The ages were determined subjectively based on the size of crown, length of spur and flexibility of the xiphoid cartilage together with information from the poultry farmers. The birds were classified as adults (cock or hen), growers (pullet or cockerel) and chicks (male and female) according to Magwisha et al (2002) and Maina (2005). Post mortem examination of chicken was carried out as described by Charlton et al (2006). Birds were skinned with all the feathers intact. The whole skin together with feathers, head and legs were preserved in 70% alcohol before further processing.
Half of the body skin was examined and the total number of parasites multiplied by two to give the overall parasite intensity. The procedure for examination of the skin for ectoparasites was carried out as described in the Ministry of agriculture food and fisheries (1986) manual.
The ectoparasites were dehydrated first in 80%, then 90% and finally 100% alcohol before being cleared in xylene and mounted on a slide for final identification with light microscope. They were identified according to their morphological characteristics using entomological keys of Soulsby (1982), MAFF (1986), Wall and Shearer (1997) and Arends (2003).
The ticks were identified using tick identification keys available from Ruedisueli and Manship, (2006) and Walker et al (2003). Important morphological features used in tick identification included size of the tick, position of head and mouthparts (capitulum) relative to the thorax and abdomen on dorsal and ventral views and shape of the body.
Infestation with Cnemidocoptes mutans was classified on a clinical evaluation based on the presence of hypertrophic dermatitis on the legs as follows: + = no macroscopic changes, no visible sign of the mite infestation though mites were present on laboratory examination; ++ = minor scale formation only the distal parts of the legs; and +++ = massive hypertrophic dermatitis with involvement of the whole leg. Scrapings were digested with potassium hydroxide and used to identify the developmental stages and adult parasites (Permin et al 2002).
Data from the study were entered in Ms-Excel, and later exported to Genstat® Discovery edition 3 for descriptive statistic analysis. To test differences in parasite-specific prevalence between the three chicken age groups, two sex groups and two agro-ecological zones, the two sample binomial test (Genstat® Discovery Edition 3) was used.
The prevalence of parasites was defined as the total number of birds infested with a particular parasite group/ species divided by the number of chicken examined at a point in time (Margolis et al 1982). A Kruskal Wallis one-way analysis of variance (Genstat® Discovery Edition 3) was used to analyze the influence of three age and two sex groups and two agro-ecological zones on the prevalence and parasite burden (intensity). A critical probability of 0.05 was adopted throughout as a cut-off point for statistical significance between groups compared.
One thirty eight chickens (95.8%) had one or more types of ectoparasites, namely; lice, mites, fleas and soft ticks (which lice, mites, fleas & soft ticks were observed in this study?). One thirty one birds had lice, 107 mites, 42 sticktight fleas and 8 had soft ticks. Of the 138 infested birds, 25 had single while 113 had mixed infestations in the following combinations in descending order: lice and mites (49.3%); lice, fleas and mites (18.8%); lice and fleas (6.5%); lice, mites and ticks (2.9%); lice, fleas, ticks and mites (2.9%); and fleas and mites (1.5%); (Table 1).
|
Table 1. Ectoparasite groups (single or mixed infestations), number of birds infested and percentage prevalence parasitic rate in indigenous village chicken |
||
|
Ectoparasite groups |
Number of birds in the group |
Percentage parasitic prevalence rate (x/144) |
|
Lice |
20 |
14.5 |
|
Fleas |
2 |
1.5 |
|
Ticks |
0 |
0.0 |
|
Mites |
3 |
2.2 |
|
Lice and Fleas |
9 |
6.5 |
|
Lice and Mites |
68 |
49.3 |
|
Fleas and Mites |
2 |
1.5 |
|
Lice, Fleas and Mites |
26 |
18.8 |
|
Lice, Mites and Ticks |
4 |
2.9 |
|
Lice, Fleas, Ticks and Mites |
4 |
2.9 |
|
Total prevalence |
138 |
95.8 |
Lice were the most prevalent parasites (90.0%). In species specific prevalence, the prevalence was as follows in descending order: Menopon gallinae (97.7%), Epidermoptes species (83.2%), Cnemidocoptes mutans (66.4%) and Lipeurus caponis (63.4%). The study documents Epidermoptes species, Laminosioptes cysticola and Megninia species for the first time in Africa as well as Lipeurus caponis and Goniodes gigas in Kenya (Table 2).
| Table 2. Prevalence rates of various ectoparasitic species found on indigenous chicken and their predilection sites | |||
|
Ectoparasite |
Common predilection site |
Number of birds with parasite |
Percentage prevalence |
|
Lice |
|
131/144 |
90.0 |
|
1. Menopon gallinae |
Feather shafts and all over the body |
128 |
97.7 |
|
2. Lipeurus caponis |
Underside of the large wing feathers |
83 |
63.4 |
|
3.Goniodes gigas |
Body feathers |
41 |
31.1 |
|
Mites |
|
107/144 |
74.3 |
|
4. Cnemidocoptes mutans |
Lower limbs (non-feathered areas) |
71 |
66.4 |
|
5. Dermanyssus gallinae |
Entire body of bird |
8 |
7.5 |
|
6. Epidermoptes species |
On the skin of birds |
89 |
83.2 |
|
7. Laminosioptes cysticola |
On subcutaneous tissue |
2 |
1.9 |
|
8. Megninia species |
On feathers (quills) |
4 |
3.7 |
|
Stick tight flea |
|
42/144 |
29.2 |
|
9. Echidnophaga gallinacean |
Comb, wattles, eyes and around the ears |
42 |
29.2 |
|
Soft tick |
|
8/144 |
5.6 |
|
10. Argas persicus |
Ventral abdominal area and below wings. |
8 |
5.6 |
Epidermoptes species were found in 89 (83.2%) of 107 birds, which were found to be infested with mites. These mites were brownish white in color and nearly one and a half time long as broad. The opisthosoma was splitted behind to form two diverging abdominal lobes pointed at the end and bearing incrassate long and two small setae at the tip. All the tarsi were round at the tip, devoid of any claw, each bearing an ambulatory sucker with short unsegmented stalk. Their pre-tarsi were short while the sucker-like pulvillus was more funnel-shaped (Figure 1).
 |
|
Figure 1. Male Epidemoptes species obtained from a chicken from lower midland 5 showing the first pair of legs (L) which is slightly thicker and ambulatory sucker with short unsegmented stalk (AS) (magnification X40) |
Laminosioptes cysticola was found in 2 (1.9%) of the 107 birds infested with mites. This mite was characterized by long body (female mite measuring 0.3 by 0.1mm). The body had a few setae and two pairs of long setae on the posterior body margin (Figure 2).
|
|
|
Figure 2.
Laminosioptes cysticola found in chicken from lower midland zone 5
showing a long body (A) |
Megninia species was found on 4 (3.7%) of 107 chicken that were infested with mites. This mite was elongated in size. The third pair of legs was characteristically long. The legs had short pre-tarsi, while the sucker-like pulvillus was more funnel-shaped. It had tibial spurs on legs 1 and 2 and shallow incised terminal lobes (Figure 3).
|
|
|
Figure 3.
Megninia species found in a chicken obtained from lower midland 5,
showing |
Lipeurus caponis (wing louse), had an elongated, narrow body measuring about 2.2 mm in length and 0.3 mm in width. The legs were narrow and characteristically, the hind legs were about twice as long as the first two pairs. The first segment of the antennae was considerably longer than following four segments. There were characteristic small angular projections on the head in front of the antennae (Figure 4).
|
|
|
Figure 4.
Lipeurus caponis from a bird in lower midland 5 showing slender |
Goniodes gigas are large lice of about 3mm in length, brown in color and the head had concave posterior producing marked angular corners at the posterior margins. The head carried two large bristles projecting from each side of its dorsal surface. The antennae had five segments (Figure 5).
|
|
|
Figure 5.
Ventral view of Goniodes gigas from a chicken in lower highland 1
showing the antennae with five |
All adult birds were infected with ectoparasites followed by 97.7% grower and 89.6% chicks. Both male and female birds had the same prevalence (95.8%) of ectoparasites. Lower midland 5 had a slightly higher prevalence of ectoparasites (98.6%) compared to LH1 (93.1%) (p>0.05). The parasite load ranged from 10 to 178 parasites per bird, with an average mean intensity of 36.0 parasites per bird. The mean intensities of: 32.6, 12.3 and 15.6 parasites per bird for lice (overall), fleas and poultry soft ticks, respectively were recorded. Among the lice species, mean intensity was: 23.9, 9.7 and 8.6 lice per bird for Menopon gallinae, Lipeurus caponis and Goniodes gigas, respectively. Stick tight fleas (E. gallinacean) parasite load ranged from 0- 113 fleas per bird with an average of 3.8 fleas per bird, while that of poultry soft tick (Argas persicus) was 0- 28 ticks (mostly larvae stage) per bird with an average of 0.9 ticks per bird. What about fleas and soft ticks? The mean specific parasite intensity in chickens was significantly different among age groups (total lice, Menopon gallinae and Lipeurus caponis (p=0.001)) and between agro-ecological zones (total lice (p=0.016), Menopon gallinae (p=0.001) and soft ticks (p=0.004)). Ticks were only observed on chicken from Lower midland zone 5 (Mbeere) with an overall prevalence of 11.1% (p<0.05).
This study showed that ectoparasites are prevalent in indigenous chicken in Embu (LH 1) and Mbeere (LM 5) districts, at a rate of 95.8%. Similar observations of high prevalence of ectoparasites in chicken have been reported in other tropical African countries such as Nigeria (Fabiyi 1996; Sadiq et al 2003), Ethiopia (Abebe et al 1997), Zimbabwe (Permin et al 2002), Malawi (Njunga 2003); and in market birds (Maina 2005) and another district in Kenya (Mungube et al 2008). These previous studies did not focus on the influence of age and agro-ecological zones on parasite prevalence. There was also a difference in types and prevalence of ectoparasites reported. Such high prevalence could be due to the free-range system practiced in the study areas, which exposes the birds to poor hygiene on the farm and chicken houses thus, enabling them to contract a wide range of harmful ectoparasites. The free-range system provides a more sustainable environment for the parasites. Mungube et al (2008) reported that lack of control measures towards these parasites was a possible factor contributing to the high prevalence of the parasites.
Ectoparasites damage feathers, irritate and cause skin lesions, resulting in reduced performance of adult chickens and direct harm to young chicks (Arends 2003). The importance of these ectoparasites transcends the direct impact on poultry production limiting the protein available to humans. Some of these parasites encountered in this study (Argas persicus and Dermanyssus gallinae) are reported either to infest man or cause annoyance especially in rural areas where there is close association between man and domestic fowls. Argas persicus is known to infest man especially children (Arends 2003; Sadiq et al 2003).
The adult birds had a 100% overall prevalence of ectoparasite, which was slightly higher than that of growers (97.7%) and chicks (89.6%). These findings are like those reported in Zimbabwe (Permin et al 2002) and Nigeria (Biu et al 2007), in which adult birds were compared to young birds. However, comparisons of ectoparasite prevalence among the three chicken age groups (chicks, growers and adults) have not been previously reported in other tropical countries. Older birds maybe are exposed longer to the infested environment than the chicks, hence a higher prevalence rates.
Both male and female birds had the same prevalence of 95.8%. Mungube et al (2008) reported that males had a slightly higher rate of occurrence of ectoparasites compared to females, while in Nigeria, Biu et al (2007) found that female chickens had a higher prevalence of ectoparasites (15.4%), than male birds (14.7%). In both studies, the differences were not significant statistically. May be the difference is due to locality and other factors. There are conflicting reports on the impact of host sex on prevalence of avian lice. It has been stated that lice are more prevalent on male birds in case of sparrows (Woodman and Dicke 1954). However, no difference in louse prevalence with respect to host sex has been noted in domestic hens (in case of Menopon gallinae) (Saxena et al 1995). During the present studies, sex related differences in the prevalence rate were not found to be significant. Thus, it may be stated that some host factors may occasionally cause variation in louse prevalence in some cases (Saxena et al 1995), but generally there is no significant difference in prevalence with respect to host sex. These findings suggest that sex is not an influential factor on the prevalence rates of ectoparasites in poultry.
Lower midland zone 5 was found to have a slightly higher prevalence rate (98.6%) compared to LH1 (93.1%). There was a significant difference in intensities of lice (overall) and Menopon gallinae between the agro-ecological zones, with LM5 having a higher intensity than LH1, probably due to agro-climatic variation. Lower midland 5 is a semi-arid area with lots of shrubs, high temperatures and lower altitude. This provides a suitable ecological environment for productive parasite lifecycles. Warmer temperatures lead to higher parasite prevalence and/ or intensities (Fabiyi 1996). However, LH1, in Embu district is a wet agricultural area with high rainfall, high altitude and lower temperatures. Although these conditions interfere with parasite life cycles, the vegetation is usually dense to provide amble breeding place for these ectoparasites. Also, due to cold weather experienced in LH1 agro-ecological zone, this tends to modify the behavior of chicken by encouraging confinement, which in turn facilitates the transfer of parasites from infested to non-infested chicken. This may explain the slight difference in prevalence rates between the agro-ecological zones despite their varying agro-climatic conditions.
We acknowledge the chairman and technical staff of the Department of Veterinary Pathology, Microbiology and Parasitology, Kabete for their assistance during the setting up and running of laboratory work. Local guides and smallholder farmers from various villages in Embu and Mbeere where the study birds were purchased are appreciated for their indefatigable support and cooperation while sourcing for the study material. The study was financed by Danish International Development Agency (DANIDA) through the ENRECA program “Productivity and Health of Smallholder Livestock in East Africa (PHSL) and University of Nairobi grant no. 500-661-202.
Abebe W, Asfaw T, Genete B, Kassa B and Dorchies P H 1997 Comparative studies of external parasites and gastrointestinal helminthes of chicken kept under different management systems in and around Addis Ababa (Ethiopia) Review of Medicine and Veterinary 148: 497-500.
Arends J J 2003 External parasites and, poultry pests. In: Diseases of poultry. 11th edition. Edited by Calnek W B, John H, Beard W C, McDougald L R and Saif Y M Iowa State Press, Blackwell Publishing Company, Ames, Iowa. P 905- 930.
Biu A A, Agbede R I and Peace P 2007 Studies on ectoparasites of poultry in Maiduguri, Nigeria. Nigerian Journal of Parasitology 28: 69-72
Charlton B R, Bermudez A J, Boulianne M, Halvoson D A, Schrader J S, Newman L J, Sander J E and Wakenell P S 2006 Necropsy of the fowl. In, Avian disease manual. 6th edition. American Association of avian Pathologist. Athens, Georgia. P 232-233.
Fabiyi J P 1996 Association between duration of humid season and geographical distribution of patterns of different species of chewing lice (Mallophaga: Insecta) infesting domestic chickens in Nigeria. Journal of Parasitology 82: 1034-1036.
Kiptarus J K 2005 Focus on Livestock sector: Supply policy framework strategies, status and links with value addition. Workshop on value Assess food and export investment. Held at Grand Regency hotel, Nairobi, Kenya. 3rd March 2005.
Magwisha H B, Kassuku A A, Kvysgaard N C and Permin A 2002 A comparison of the prevalence and burdens of helminth infections in growers and adult free-range chickens Tropical Animal Health and Production 34: 205-214.
Maina A N 2005 Prevalence, intensity and lesion associated with gastrointestinal parasites of indigenous chicken in Kenya. MSc thesis. University of Nairobi.
Margolis L, Esch G W, Holmer J C, Kuns A M and Schad G A 1982 The use of ecological terms in parasitology. (Report of an Ad Hoc committee of American Society of Parasitologists). Journal of Parasitology 68: 131- 133.
Martin S W, Meek A H and Willberg P 1987 Veterinary Epidemiology, Principles and Methods, Iowa State University Press, Ames, Iowa. P 32
MAFF 1986 Ministry of Agriculture, Fisheries and Food (MAFF). Manual of Veterinary Parasitological Laboratory Techniques, 3rd edition, reference book 418. HMSO, London. P 118-124.
Moyer B R, Drown D M, and Clayton D H 2002 Low humidity reduces ectoparasite pressure: implications for host life history evolution. Oikos 97: 223-228
Mungube E O, Bauni S M, Muhammed L, Okwach E W, Nginyi J M and Mutuoki T K 2006 A survey of the constraints affecting the productivity of the local scavenging chickens in the Kionyweni cluster, Machakos District. KARI Katumani, Annual Report 2005.
Mungube E O, Bauni S M, Tenhagen B A, Wamae L W, Nzioka S M, Muhammed L and Nginyi J M 2008 Prevalence of parasites of the local scavenging chicken in a selected semi-arid zone of Eastern Kenya. Tropical Animal and Health Production Bulletin 40: 101-109
Njunga G R 2003 Ecto- and haemoparasites of chicken in Malawi with emphasis on the effects of the chicken louse, Menacanthus cornutus. MSc thesis. The Royal Veterinary and Agriculture University, Denmark.
Onduru D D, Gachimbi L, Maina F, Muchena F N and A Der Jager 2002 Sustaining Agricultural Production in Semi Arid area of Eastern Kenya. A case study of Mbeere District. INMASP Report No. Ke-03.
Permin A, Esmann J B, Hoj C H, Hove T and Mukatirwa S 2002 Ecto-, Endo- and Haemoparasites in free range chicken in the Gomoronzi District in Zimbabwe. Preventive Veterinary Medicine 54: 213-224.
Ruedisueli F L and Manship B 2006 Tick identification key. University of Lincoln. Available on-line at http://webpages.lincoln.ac.uk/fruedisueli/FR-webpages/parasitology/Ticks/TIK/tick-key/softticks_adult.htm. Last visited on 6th march 2008.
Sadiq N A, Adejinmi J O, Adedokun O A, Fashanu S O, Alimi A A and Sofunmade Y T 2003 Ectoparasites and haemoparasites of indigenous chicken (Gallus domesticus) in Ibadan and environs. Tropical Veterinarian 21: 187-191.
Saxena A K, Kumar A, Surman and Singh S K 1995 Prevalence of Menopon gallinae Linne. (Phthiraptera : Amblycera) on poultry birds of Garhwal. Journal of Parasitic Diseases 19: 69-72.
Soulsby E J L 1982 Helminths, Arthropods and Protozoa of Domestic Animals. 7th Edition. London: Bailliere and Tindall, East Sussex, UK.
Walker A R, Bonattour A, Camicas J J, Estrada-Pena, Harok I G, Latif A A, Pegram R G and Preston P M 2003 Ticks of Domestic animals in Africa: A guide to identification of species. http://www.alanrwalker.com/index/cms-filesystem-action/tickguide-africa-web-08.pdf
Wall R and Shearer D 1997 Veterinary entomology. 1stedition. Chapman and Hall, London. P 43-95.
Woodman W L and Dicke R J 1954 Population fluctuation of the mallophagan parasite Brueelia vulgata (Kellogg) upon the sparrow. Transactions of the Wisconsin Academy of Sciences, Arts and Letters 43: 133-135.
Received 26 May 2010; Accepted 22 July 2010; Published 1 November 2010