
| Livestock Research for Rural Development 31 (6) 2019 | Guide for preparation of papers | LRRD Newsletter | Citation of this paper |
The integration of freshwater prawn (Macrobrachium rosenbergii) and red seaweed (Gracilaria tenuistipitata) was conducted consisting of five treatments and run in triplicate. Prawn monoculture fed 100% commercial feed ration as a control, other four treatments prawn were integrated with red seaweed (1.5 kg/m3) and received 80%, 60%, 40% and 20% feeding rations. Prawn juvenile (0.53±0.04 g) were stocked in the 250-L tanks with density of 100 ind./m3 and at salinity of 5 g/L for 90 days.
Results showed that the concentrations of TAN and NO2- in the integrated tanks were lower than that in the monoculture. Survival and growth rates of prawn in the integrated treatments received 40% feed ration were superior to those in the control and other treatments, and feed conversion ratio could be reduced up to 66.7% compared with the control group. Additionally, ash, protein and lipid contents of prawn meat in co-culture were better or similar to that of the control. It can be suggested that applying 40% commercial feed ration in the integration of M. rosenbergii and G. tenuistipitata gave optimal growth performance, improved survival and feed efficiency, as well as maintain better water quality in prawn culture.
Key words: feed conversion ratio, growth, survival, water quality
The giant freshwater prawn Macrobrachium rosenbergii de Man, 1879 is one of the most important freshwater crustacean species in the world and has been introduced into more than 40 countries, of which the major producing countries are in Asia (New and Nair 2012). According to Chand et al (2015), the prawn grew and survived satisfactorily at salinity range of 0–15 ppt, implying that the species can be cultured commercially in brackish water region. In the Mekong Delta of Vietnam, M. rosenbergii is one of the target aquaculture species, typically the alternative rice-prawn farming system. This activity has recently expanded rapidly and largely to the brackish water area (Hai et al 2017).
Red seaweed Gracilaria species are euryhaline, rapid growth and having high nutrient uptake (nitrogen and phosphorus) as they have been commonly used as biofilter in mariculture ecosystems to improve the water environment (Peng et al 2009; Yang et al 2015) and served as supplemental food in the integrated system in shrimp farming that offered many beneficial effects to the culture system (Andayani et al 2016). Co-culturing the black tiger shrimp (P. monodon) with G. tenuistipitata reduced the concentrations of nitrogen and phosphorus in the culture tank and feed supply for shrimp (Anh et al 2018). Recently, G. tenuistipitata has been found abundantly in the improved extensive shrimp farms in Mekong delta and considered to be good for shrimps. Currently, Mekong delta has been affected by climate change, more and more coastal areas of this area are going to be vulnerable to saline water intrusion. Freshwater prawns are excellent options for integrated systems in brackish water bodies since they are omnivores and can therefore take advantage of a wide range of feed residuals, either from aquatic or terrestrial species. Besides, due to their benthic habit, they have a well-defined spatial distribution in the environment, thus favoring interaction with various species of fish, other animals, and even with plants (Marques et al 2016). The present study aims to find out the appropriate commercial feed ration in the integrated system of the M. rosenbergii and G. tenuistipitata to obtain optimal growth and feed efficiency in prawn culture. This finding can provide scientific basis for further research in field conditions and development of this model in the Mekong delta of Vietnam and other countries.
Freshwater prawn juveniles were provided from the private hatchery in Can Tho city, Vietnam and acclimated in a 2 m3 tank for 2 days prior to the start of the experiment. Red seaweed (G.tenuistipitata) was collected from the improved extensive farm in Bac Lieu province, and then red seaweed was separated from other seaweeds and acclimated to adapt experimental salinity (5 g/L) for 5 days. Commercial pellet feed (Grobest) was produced by Grobest Company, Dong Nai province, Vietnam. Proximate composition of red seaweed and commercial feed are shown in Table 1.
| Table 1. Proximate composition of G. tenuistipitata (% of dry matter) and pellet feed | |||||
| Feed type | Moisture | Protein | Lipid | Ash | Fiber |
| G. tenuistipitata | 85.4 | 18.34 | 1.36 | 28.47 | 10.26 |
| Grobestfeed* | £11 | ³42 | 5-7 | £15 | £3 |
| * Information based on label from Growbest Company | |||||
The freshwater prawn (M. rosenbergii) integrated with red seaweed (G. tenuistipitata) received different feeding rations consisted of five treatments. Shrimp monoculture fed 100% commercial feed ration as a control (F100), and shrimp co-culture received 80%, 60%, 40% and 20% feed rations. Treatments are abbreviated as GF80, GF60, GF40 and GF20, respectively. The experiment followed a completely randomized design in triplicate tanks for 90 days.
The experimental system was placed under a transparent roof at the College of Aquaculture and Fisheries, Can Tho University, Vietnam. All culture tanks were maintained continuous aeration and exposed to a natural photoperiod (light intensity fluctuated from 2,500 to 18,500 lux during daytime). Water temperature, pH and alkalinity in the culture tanks varied in the ranges of 26.2-29.4°C, 7.5- 8.1 and 72-108 mgCaCO3/L, respectively. Prawn juveniles with mean initial weight of 0.53±0.04 g and total length of 3.31±0.26 cm were stocked in fifteen 250-L of plastic tanks, filled with 200-L seawater at salinity of 5 g/L. Stocking density was 100 prawns/m3 (20 prawns/tank) for all treatments. Red seaweed was added to the integrated tanks at a biomass of 1.5 kg/m3 (0.3 kg/tank).
 |
| Photo 1. (A) Experimental system, (B) Integrated tank, (C) Freshwater prawn at harvest |
A commercial feed (Growbest) was distributed four times a day (7:00, 11:00, 16:00 and 21:00 h) on the feeding trays; 100% feed ration (F100) was equivalent to a daily ration of 5% prawn biomass. The amount of feed supplied in this treatment were strictly managed and adjusted based on the absence or presence of feed after one hour of feeding and shrimp weight. Other daily feed ration treatments, the amount of feed required was subtracted from the determined amount of the F100 treatment. Faecal matter and excretory products were siphoned out every two days and refilled with new water to the initial volume. Water exchange was done every 15 days and about 50% of the tank volume during culture period.
Sampling was conducted at 10-day intervals for water quality, biomass of red seaweed and prawn weight during experimental period. The concentrations of TAN (total ammonium nitrogen) and NO2- were determined using a standard method of APHA (1998). Water samples were taken in the culture tanks prior to water exchange. Ten prawns were randomly collected from each tank, weighed in group on a digital Mettler scale and the mean weight was determined. These prawns were returned to the original tanks. Seaweed biomass in each tank was collected, excess water in harvested seaweed biomass was removed with tissue paper, and the wet weight was recorded with a balance. At the end of the experiment, final weight and length of prawn were measured individually and counted to calculate survival, and proximate of prawn meat was also evaluated. Growth data of experimental prawn consisting of weight gain (WG), daily weight gain (DWG), specific growth rate (SGR) and survival; feed conversion ratio (FCR) were calculated using the following equations:
Weight gain (g) = Final weight - Initial weightDWG (g/day) = (final weight - initial weight)/cultured days x100
SGR (%/day) = ((ln final weight) – (ln initial weight))/cultured days x100
Survival (%) = Final number of prawn/Initial number of prawn x 100
FCR = Feed provided (dry weight)/Weight gain (wet weight)
Proximate analysis (moisture, crude protein, lipid, fiber and ash) of red seaweed and prawn meat were carried out according to the standard methods of AOAC (2000).
The percentage values were normalized through arcsine transformation before statistical analysis. For all treatments, results were analyzed statistically with one-way ANOVA analysis of variance to find the overall effect of the treatment (SPSS, version 16.0). Turkey test was used to identify significant differences between the mean values at a significant level of p<0.05.
Variations in the concentrations of TAN and NO2- in the culture tanks are presented in Figure 1. For the integrated treatments, the concentrations of TAN and NO2- decreased with decreasing feeding rations, the 80% feeding ration treatment (GF80) reached the highest levels among integrated treatments (0.47 mg/L and 0.71 mg/L for TAN and NO2-, respectively). For the monoculture tanks, the average contents of TAN and NO2- at day 10 were 0.23 mg/L and 0.56 mg/L respectively and tended to increase with the culture period. At the end of the experiment, these values were 1.53 mg/L and 1.91 mg/L which were 3.3 and 2.7 times higher compared to that of the GF80 treatment for TAN and NO2-, correspondingly. These results indicated that the presence of red seaweed combined with reduction of feeding rates that greatly reduced the amount of nitrogen while improving water quality in the integrated tanks.
 |
| Figure 1. Fluctuation of TAN and NO2- concentrations during the experimental period |
Previous studies found that Gracilaria species have been effectively applied as a bio-filter in aquaculture systems, as they can absorb the ammonia, nitrate and phosphate for their growth that in turn to improve water quality (Susilowati et al 2014; Samocha et al 2015; Yang et al 2015). Similar finding were reported by Andayani et al (2016), the integration of G. verrucosa as a biofilter agent in Litopenaeus vannamei shrimp farming had a positive impact on water quality. The results of present study are in agreement with the study of Anh et al (2018), the contents of TAN and NO2- in co-culture of black tiger shrimp Penaeus monodon with G. tenuistipitata were significantly lower than in monoculture. In this experiment, the concentrations of nitrogen compound in monoculture was much higher than that in co-culture, these values remained within acceptable limits for growth of freshwater prawn (Marques et al 2016).
 |
| Figure 2. Variation in biomass of red seaweed G. tenuistipitata during the experiment |
Biomass of red seaweed G. tenuistipitata had a tendency to increase until day 45 and then gradually declined with culture duration (Figure 2). In this study, prawn juveniles received commercial feed at low rations and consumed certain amounts of red seaweed present in the culture tanks as supplemental food that can be one of the reason causing reduction of seaweed biomass. Additionally, the presence of seaweed in shrimp culture tanks not only improved water quality and supplemental food, but also served as a shelter that helped enhance the survival and production of shrimp (Izzati 2011; Andayani et al 2016).
After 90 days of culture, the final weight of prawn varied from 7.60 to 10.21 g, of which the smallest and largest animals were observed in the F100 treatment (monoculture with 100% feed ration) and GF40 treatment (prawn integrated red seaweed plus 40% feed ration), respectively (p<0.001). Growth rate of prawn in terms of weight gain, daily weight gain and specific growth rate followed the same pattern as observed for the final weight. Moreover, total length of prawn at harvest showed a similar effect as found for growth in weight (p<0.001).
| Table 2. Prawn performances after 90 days of culture | |||||||
| Treatment | F100 | GF80 | GF60 | GF40 | GF20 | SEM | p |
| Final weight, g | 7.60a | 9.15bc | 9.84cd | 10.21d | 8.35ab | 0.27 | <0.001 |
| Weight gain, g | 7.07a | 8.62b | 9.31bc | 9.67c | 7.61a | 0.27 | <0.001 |
| Daily weight gain, g/day | 0.078a | 0.096b | 0.103bc | 0.108c | 0.085a | 0.003 | <0.001 |
| Specific growth rate, %/day | 2.93a | 3.15bc | 3.23c | 3.26c | 3.05ab | 0.034 | <0.001 |
| Total length, cm | 8.28a | 9.72b | 10.15c | 10.23c | 9.53b | 0.19 | <0.001 |
| Survival,% | 61.7a | 81.7b | 80.0b | 81.7b | 71.7ab | 2.51 | 0.014 |
| Production, kg/m3 | 0.62a | 1.00bc | 1.06c | 1.11c | 0.80ab | 0.052 | <0.001 |
| abcd Means in the same row without common letter are different at p<0.05 | |||||||
The average prawn survival at the end of experiment ranged from 61.7% to 81.7%, in which the F100 group (control) showed the lowest value compared to other groups (p = 0.014). It was noted that integration of prawn and seaweed enhanced prawn survival, especially reducing feeding rate down to 20%, prawn survival was relatively higher than that in the control (Table 2, Figure 3).
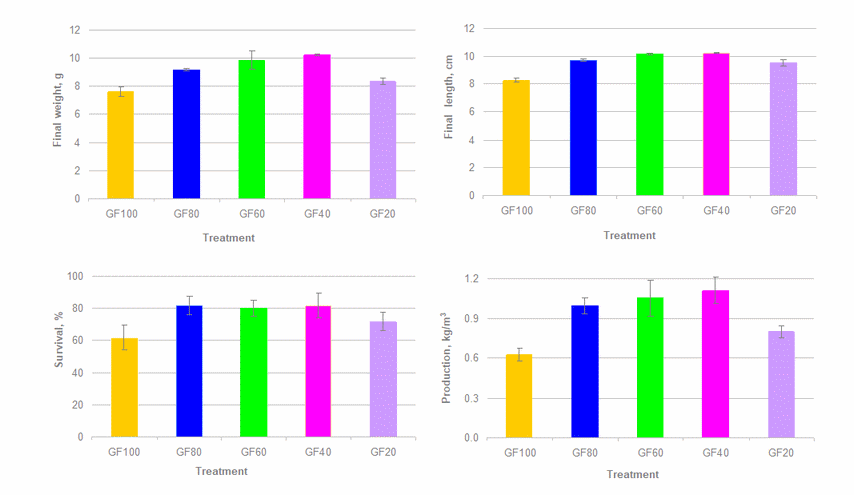 |
| Figure 3. Final weight and length, survival and production of prawn after 90 days of culture |
Several studies reported that co-culture tiger shrimp- Gracilaria, this seaweed was better in supporting the survival and growth of shrimp as seaweed served as a natural food source for shrimp (Izzati 2011). Poly-culture of L. vannamei and G. verucosa helped increase on the survival and growth rate of shrimp (Susilowati et al 2014). Our results are similar to those reported by Anh et al (2018), shrimp P. monodon co-cultured with G. tenuisitpitata attained higher survival (91.7-98.3%) compared to the monoculture (88.3%), and growth performance of shrimp received 50% and 75% feed ration were equal to or significantly higher than those in the monoculture group fed 100% feed ration. In the present experiment, the integration of prawn- red seaweed applied reduction of feed ration that indicated the shortage of feed for their requirement so shrimp could use seaweed available in the culture tanks as supplemental food. Gracilaria sp. was found to be rich in essential amino acids, fatty acids and minerals (Peng et al 2009). In this case, shrimps were lived in the good environment and good supplemental feed that favored growth of shrimps. However, prawns received 20% feed ration as they may consume more red seaweed for their diets, showed the poorest growth performance. This could be due to the differences in nutritional composition between commercial feed and red seaweed G. tenuistipitata used in this study (Table 1) where G. tenuistipitata contained low protein (18.34%) and lipid levels (1.36%) that did not meet nutritional requirement of the freshwater prawn M. rosenbergii (D'Abramo 1998).
The highest feed conversion ratio of commercial feed was observed in the control group (2.27) compared to other groups, and FCR progressively reduced with decreasing feed ration (p<0.001). The integrated system combined with reduction of feed supply from 80% to 20% feed ration, the FCR could be reduced from 26.1% to 73.4% (Table 3). Notably, the reduction of FCR in the GF40 group was 66.7% but the growth rate, survival and production of prawn were much better than those in the control group. This feeding ration could be optimal for integrating prawn M. rosenbergii-red seaweed G. tenuisitpitata. The present result was similar to the finding of Anh et al. (2018) in the case for the tiger shrimp P. monodon co-cultured with G. tenuistipitata.
| Table 3. Feed conversion ratio of prawn after 90 days of culture | |||||||
| Treatment | F100 | GF80 | GF60 | GF40 | GF20 | SEM | p |
| FCR | 2.27d | 1.68c | 1.18b | 0.76a | 0.60a | 0.16 | <0.001 |
| Reduction ratio compared to the control (%) | - | -26.1 | -47.9 | -66.7 | -73.4 | ||
The water contents (moisture) of fresh prawn meat was not different among treatments (p= 0.065). However, the contents of ash, protein and lipid were affected by the culture system and feeding ration (p<0.05). Protein content of prawn meat was highest in the GF40 treatment. The ash content showed slightly increased with reducing commercial feed ration while lipid content showed opposite tendency (Table 4).
| Table 4. Proximate composition of freshwater prawn meat (% of wet weight) | |||||||
| Treatment | F100 | GF80 | GF60 | GF40 | GF20 | SEM | p |
| Moisture | 75.7 | 75.5 | 76.1 | 75.9 | 76.3 | 0.11 | 0.065 |
| Ash | 1.35a | 1.46ab | 1.54b | 1.56b | 1.60b | 0.028 | 0.006 |
| Protein | 16.8a | 16.8ab | 16.9ab | 17.3b | 16.4a | 0.090 | 0.002 |
| Lipid | 0.59b | 0.55b | 0.52ab | 0.49ab | 0.39a | 0.021 | 0.005 |
| ab Means in the same row without a common letter are different at p<0.05 | |||||||
The author sincerely thanks Mrs. Duong Thi My Han provided freshwater prawn juveniles for conducting experiment and Tran Thanh Truong for his help during the growth trial.
Andayani S, Yuwanita R and Izzah N 2016 Biofilter application using sea weed (Gracillaria verucosa) to increase production of Vannameii shrimp in traditional pond district Bangil-Pasuruan. Res J Life Sci, 3:16–22.
Anh N T N, Ngan L T H, Vinh N H and Hai T N 2018 Co-culture of red seaweed (Gracilaria tenuistipitata) and black tiger shrimp (Penaeus monodon) with different feeding rations. IJSRP, 8:269–277.
AOAC 1990 Official methods of analysis. Association of Official Analytical Chemists, Arlington, Virginia, 15thedition.
APHA 1998 Standard methods for the examination of water and wastewater. The Association, Washington, DC.
Chanda B K, Trivedi R K, Dubey S K, Rout S K, Bega M M and Das U K 2015 Effect of salinity on survival and growth of giant fresh water prawn Macrobrachium rosenbergii (de Man). Aquaculture Reports, 2:26-33.
D'Abramo L K 1998 Nutritional requirements of the freshwater prawn Macrobrachium rosenbergii: comparisons with species of Penaeid shrimp. Reviews in Fisheries Science, 6:153-163.
Devi D V P, Hareesh K and Reddy M S 2015 Studies on the proximate composition of tropical freshwater prawn Macrobrachium rosenbergii. International Journal of Fisheries and Aquatic Studies, 3: 329-336.
Hai T N, Huong H K, Viet LQ, Huong D T T and Phuong N T 2017 Giant freshwater prawn (Macrobrachium rosenbergii de Man, 1879) farming in brackish water areas of the Mekong Delta, Vietnam. Can Tho University Journal of Science, 7:82-90.
Izzati M 2011 The role of seaweeds Sargassum polycistum and Gracilaria verrucosa on growth performance and biomass production of tiger shrimp (Penaeous Monodon Fabr). Journal of Coastal Development, 14: 235-241.
Marques H L A, New M B, Boock M V, Barros H P, Mallasen M and Valenti W C 2016 Integrated freshwater prawn farming: State-of-the-art and future potential. Reviews in Fisheries Science & Aquaculture, 24:264-293.
New M B and Nair C M 2012 Global scale of freshwater prawn farming. Aquaculture Research, 43:960-969.
Peng C, Hong-Bo S, Di X and Song Q 2009 Progress in Gracilaria Biology and Developmental Utilization: Main Issues and Prospective. Rev Fish Sci, 17: 494–504.
Reddy S K V, Babua K R and Raju M R 2013 Proximate composition of the prawn, Macrobrachium rosenbergii from Andhra Pradesh coast, India. INT J CURR SCI, 8: E 16-20.
Samocha T M, Fricker J, Ali A M, Shpigel M and Neori A 2015 Growth and nutrient uptake of the macroalga Gracilaria tikvahiae cultured with the shrimp Litopenaeus vannamei in an Integrated Multi-Trophic Aquaculture (IMTA) system. Aquaculture, 446:263-271.
Susilowati T, Hutabarat J, Anggoro S and Zainuri M 2014 The improvement of the survival, growth of naname shrimp (Litopenaeus vannamei) and seaweed (Gracilaria verucosa) based on polyculture cultivation. IJMARCC, 1:6–11.
Yang Y, Chai Z, Wang Q, Chen W, He Z and Jiang S 2015 Cultivation of seaweed Gracilaria in Chinese coastal waters and its contribution to environmental improvements. Algal Research 9:236–244.
Received 28 March 2019; Accepted 2 April 2019; Published 4 June 2019