
| Livestock Research for Rural Development 28 (9) 2016 | Guide for preparation of papers | LRRD Newsletter | Citation of this paper |
The temperament of Guzerá females was evaluated to identify genetic and non-genetic factors influencing the expression of this trait, providing information to support decision-making in breeding programs. Temperament assessed by reactivity was evaluated in a livestock scale using visual scoring (TS) and an electronic device (TR). Measurements were repeated during the rainy and dry seasons in 1354 females from five herds comprising 2663 records. Estimates of (co)variance components and genetic parameters were obtained using the derivative-free restricted maximum likelihood method. The reactivity of Guzerá females widely varied. There is indication that the bulls used in these herds have been chosen based also on their daughters’ temperament.
The effects of herd, season, physiological state, weight class, entry order class, and age at time of measurement were significant (p< 0.001) sources of variation in TR. Herd was the most important factor associated with the reactivity and particularly handling practices are likely to explain the differences. The heritabilities of TS and TR were estimated as, 0.29 ± 0.08 and 0.06 ± 0.02, respectively. The genetic correlations were 0.89 between TS and TR and -0.46 between TR and test-day milk production. This study suggested that variation in TR is mainly influenced by non-genetic effects. Genetic parameter estimates still do not allow for an accurate establishment of the potential of TR for selection. Further studies using larger datasets should be performed to provide additional evaluation of female temperament.
Keywords: animal behaviour, animal handling, dairy cattle, reactivity, Zebu
Studies on animal behaviour are important, particularly considering its close relationship to welfare, animal performance, product quality and profitability, and, more recently, its association to consumer acceptability of animal production systems with legal and market implications (Bowles et al 2005; Von Keyserlingk et al 2009).
Temperament is a behaviour trait of major interest in animal science (Sewalen et al 2010). Cattle temperament is defined as the animal’s behavioral response to handling by humans (Burrow, 1997). This trait also depends on aspects inherent to animal, animal-animal and animal-environment interactions (Munksgaard et al 2001; Adamczyk et al 2013). Therefore, there are several methods to assess this trait (Fraser, 2009; Hoppe et al 2010; Sebastian et al 2011; Schwarztzkopf-Genswein et al 2012).
The mechanisms underlying the cattle temperament are determined by genetic and non-genetic factors (Goddard et al 2000). Genetic parameter estimates have ranged widely (Adamczyk et al 2013). In turn, the influence of routine management, prior experience and increasing age is very important (Boivin et al 1998; Grandin, 2000; Tözsér et al 2003; Petherick et al 2009).
However, the experimentation to accurately predict these parameters is often viewed as costly and time consuming, indicating more research is required to understand the expression of temperament and establish the potential of this trait for breeding purposes in cattle (Gutiérrez-Gil et al 2008; Adamczyk et al 2013). The majority of these studies have been carried out with beef cattle (Barrozo et al 2012; Sant’Anna and Paranhos da Costa 2013; Rueda et al 2015). To our knowledge, the few studies with dairy cattle has been undertaken particularly with the Holstein breed (Bos taurus) (Sutherland et al 2012; Friedrich et al 2015).
The Guzerá breed is an important genetic resource for tropical livestock production broadly used in Brazil because of their adaptability to harsh environments and low input systems (Peixoto et al 2010). Although competitive, Guzerá temperament is considered a limiting factor, aspect that can result in low productive performance and product quality, handling difficulties, and damage to facilities (Paranhos da Costa and Broom 2001; King et al 2006; Petherick et al 2009; Sebastian et al 2011).
In view of the importance of the Guzerá to tropical production, the objectives of this study were to quantify the temperament of Guzerá females, the factors influencing it, and the association between temperament and milk production.
Data from 1354 Guzerá females, aged 6 to 295 months, from different categories (heifers to multiparous) and physiological states (lactating and non-lactating) were recorded from five herds (Table 2). The herds are located in the Brazilian Southeastern Atlantic Rainforest and Savannah-like biomes, being representative of the Guzerá breed in Brazil, and the main suppliers of proven genetic for pure and crossbred cattle.
Farms used extensive pasture-based systems with roughage supplementation offered during the dry season and a concentrated diet provided in the milking parlour throughout lactation. A salt-based mineral supplement was offered daily in paddock. Futher details on each farm are presented below:
A –The farm area has an extension of 496 ha, and the manager is an experienced technician. Roughage was supplemented to all animals in the dry period. Primiparous were milked twice a day, and multiparous once a day. Calves are kept with their mothers along the afternoon
B – The farm area has an extension of 2300 ha. Roughage was supplemented only to lactating cows. Concentrate was offered also to cows during transition. Calves were tamed.
C – The farm area has an extension of 1166 ha. Silage composed of sugar-cane plus urea was supplemented to all animals. Cows were manually milked twice a day.
D – The farm area has an extension of 100 ha. Roughage and concentrated supplementation were offered to all animals. High-protein concentrates were regularly offered to calves until they were two years old. The calves were handled often after weaning.
E – The farm area has an extension of 1183 ha. Roughage supplementation was provided to all animals. Concentrate supplementation was only provided to first-lactating cows. Calves were fed a high-protein concentrate only during the dry period. The maternity paddock was located in a humid area. Calves were kept with cows until sixty days before the following calving.
The beginning of reproductive life was based on the live weight or body condition score. Artificial insemination was usually concentrated in the breeding season. Cows that fail after two attempts to conceive are submitted to natural service. Cows were taken to maternity in the transition period of thirty days before calving. During the transition, the maternity was visited twice a day by a stockman to observe for signs of delivery. In the majority of the herds, excepted for herd E, pregnant heifers were taken to the milking parlour daily during this period where they were subjected to same routine practices as the lactating cows, except milking.
Lactating cows were managed in paddocks near the milking parlour, while the heifers and dry cows remained in more distant paddocks. Lactating cows were joined to their calves before the milking and milked manually in the presence of the calf. After milking, cows were usually kept with their calves in the afternoon and then were separated for the night. Weaning usually occurred as a result of reduced milk production (4-5 kg) or at 305 days in lactation, when the cows were dried-off.
Management practices varied in some aspects from one herd to another. To give support to discussion and conclusions, common details were presented in Table 1. Particularities of each herd are described just below.
|
Table 1. Management practices adopted in the Guzerá herds of this study. |
|
|
Practices |
Herds |
|
Presence of the owner during handling |
B,C,E |
|
Agricultural technician assistence |
A,B,D |
|
Shading in the paddocks |
C,D |
|
Frequent stockmen supervision and training |
A,C |
|
Calves are kept with cows more than 48 h after calving |
A,D |
|
Use of artificial insemination |
A,C,D,E |
|
Use of timed artificial insemination |
B,C,D |
|
Training of pregnant heifers in the milking parlor during transition |
A,B,C,D |
|
High-protein concentrate available to pre-weaning calves and lactating cows |
A,B,C |
|
Fixed amount of concentrate supplementation of lactating cows |
A,B,D |
|
Culling of bad temperament females |
A,C,D |
|
No use of bulls whose daughters are of bad temperament |
A,C |
|
No use of stingers in animal’s management |
A,B,C,D |
Measurements of temperament were performed for one year. They consisted of rainy season measurements (n = 1354) that were repeated in the dry season (n = 1324). Not all females that were evaluated during the rainy season were evaluated during the dry season because of sales or disposals. The average air temperature recorded in the localities nearest the herds was lower for the dry season (17ºC) than that for the rainy season (28ºC), and the relative humidity ranged from 50 to 75% and 60 to 85% for the dry and rainy seasons, respectively.
The temperament of females were evaluated under restraint in a confinement weighing scale used occasionally in routine veterinary inspections and practices, and also using subjective indicators. Both of them based on the general activity of an individual (Réale et al 2007)
On the test-day, the females were brought to the corral in groups according to the herd’s standard management practices and established based on age and physiological state (lactating and dry). Only the stockmen managed the animals while the measurements were obtained to minimise stress induced by the presence of strangers. Reactivity was assessed while the animals were restrained in the weighing scale whose dimensions (height X length X width) averaged 2.0 X 2.8 X 1.0 meters. Also, the order in which animals entered the scale was recorded.
Two indicators, subjective visual scores (TS), and the objective test of reactivity (TR) were used for temperament assessment, and recorded while the animals were confined in scales. For TS, temperament was subjectively rated by the same two trained technicians on a grading scale based on the frequency and vigour of the movement of the animals’ heads, legs and tails, and vocalisation episodes inside the scale. This scores ranged from 1 to 5, with grade (1) meaning “calm, standing, no movement, with occasional and mild tail movements”, (2) for “calm, few movements, with occasional and mild tail movements”, (3) for “restless, with frequent and vigorous tail movements, and audible breathing”, (4) for “vigorous and sudden movements, with frequent and vigorous tail movements, and audible breathing but without vocalisation”, and (5) for “frenzy, vigorous and continuous movements, vigorous and continuous tail movements, jumping, jerking, vocalising, and snorting”.
Additional score was contributed by the stockman who routinely handled the animals reflecting thus a long term impression of how females usually behave. To reduce subjectivity, the mean values obtained by each examiner, including that by stockman, were calculated and used as the final score. The reactivity was also measured objectively using an electronic device attached to the scale. This device was equipped with an accelerometer that measures the frequency, intensity and temporary variation in animal movements for 20 seconds from the time the animal enters the scale (Bergmann et al 2010). Total pulse counts were used as a variable to evaluate animal reactivity while in the containment weighing scale according to a continuous grading scale. Data was automatically processed on specific software.
To present the results comparatively to TS, TR (as 10-log reactivity) was grouped in five categories (<2.6; ≥ 2.6 − < 2.98; ≥ 2.98 − < 3.36; ≥ 3.36 − < 3.74; ≥ 3.74). After analysis of data consistency, the distributions of the new trait (TR) and the TS were evaluated using a test for normality. Descriptive analyses were performed to summarise the data and verify relationships between some variables (herd, season, physiological status, entry order to measurement, and age), using the procedures available in SAS (Statistical Analysis System, version 9.2).
The number of data for temperament in each herd, physiological state, entry order, weight and age category used in the variance analysis model is described in table 2. TR values were compared between the rainy and dry seasons using Pearson’s correlation coefficient and Bartlett’s test for homogeneity of variance. Pearson’s correlation test was also used to evaluate the association between reactivity data and the age in months.
|
Table 2. Description of data set used in the variance analysis according to the categories of each effect tested. |
||||
|
Effects |
Categories |
Rainy |
Dry |
Total |
|
Herd |
A |
92 |
87 |
179 |
|
B |
445 |
451 |
896 |
|
|
C |
341 |
328 |
669 |
|
|
D |
110 |
107 |
217 |
|
|
E |
299 |
310 |
609 |
|
|
Physioliogical stage |
Lactating |
940 |
891 |
1831 |
|
Non-lactating |
347 |
392 |
739 |
|
|
Entry order |
1 |
707 |
814 |
1521 |
|
2 |
292 |
349 |
641 |
|
|
3 |
288 |
120 |
408 |
|
|
Weight |
1 |
371 |
292 |
663 |
|
2 |
326 |
328 |
654 |
|
|
3 |
317 |
314 |
631 |
|
|
4 |
273 |
349 |
622 |
|
|
Age |
1 |
161 |
134 |
295 |
|
2 |
423 |
399 |
822 |
|
|
3 |
354 |
382 |
736 |
|
|
4 |
302 |
327 |
629 |
|
| N*: Number of females in each season | ||||
The analysis of variance model considering repeated measures in each season was chosen because it provided the best fit based on the Akaike Information Criterion (AIC; Akaike, 1974; Dobson, 2002) and the Bayesian Information Criterion (BIC; Schwarz 1978; Burnham and Anderson 2003) values, and the likelihood ratio test. A total of 2505 observations from cows with data in both the rainy and dry periods were used in the analysis of variance.
The effects studied included herd (A, B, C, D, and E), season (rainy or dry), physiological state (non-lactating or lactating), weight class (≤ 408 kg; ≥ 409 − ≤ 462 kg; ≥ 463 − ≤ 514 kg; > 514 kg), class of entry order into the livestock scale (first 30; 31 to 60; after 60), and of age at time of measurement (≤ 24 months; > 24 − ≤ 48 months; > 48 − ≤ 72 months; > 72 months). In substitution, age was also analysed as a covariate to obtain regression coefficients (linear and quadratic) to plot temperament trend according to the age. To present results for fixed effects, a simpler model was used. The methodology to access genetic variance and parameters is presented ahead. The following models to analyse reactivity were:
Yijklmn = µ + Hi + Sj + PSk + Wl + EOm + An + εijklmn
where Yijklmn= reactivity of the ijklmn female, Hi = fixed effect of herd i,Sj = fixed effect of season j, PSk = fixed effect of physiological state k , Wl = fixed effect of weight l, EOm = fixed effect of weighing scale m, An = fixed effect of age female n or, in substitution, the termb1(Iijklmn – I) + b2(Iijklmn – I)2 was included considering age as a covariate, withb1 and b2 = linear and quadratic regression coefficients, Iijklmn = age of female ijklmn at measurement, I = average age of females at measurement, and εijklmno = random residual term for observation n.
The restricted maximum likelihood method available in the MIXED procedure, which fits generalised linear mixed (GLM) models to input data using the restricted maximum likelihood method to estimate (co)variances was used for detailed variance analysis.
(Co)variance components and genetic parameters were estimated using the
restricted maximum likelihood method under a uni and bivariate repeatability
linear animal model, applying the derivative free algorithms available in
the MTDFREML software (Boldman et al 1995). The general model can be written
in matrix form as follows:

where y is a vector of observations for temperament (reactivity, visual score or both; reactivity and test-day milk production), X is the incidence matrix of fixed effects, β is the vector of fixed effects including the categories herd, season, physiological state, and the effect of age of female at the time of measurement as covariate (linear and quadratic effects), Z is the incidence matrix for random additive genetic effects of animal (a) and permanent environment effects (p); u is the vector of random additive genetic (a) and permanent environment (p) effects, and e is a vector of random residual term. For random elements, the following distributions were assumed: a ~ N(0,Aσ2 a), where A = numerator relationship matrix, p ~ N(0,Iσ2p), and e ~ N(0,Iσ2e).
A total of 2648 reactivity records, from 1324 females, was used for analysis, and the relationship matrix contained 12949 animals.
Sly animals tended to enter the scale at the end of measurement, thus entry order into the livestock scale was not included in the model for genetic parameter estimation because it is considered an indicator of temperament per se and could be reflecting the same trait (Maffei et al 2006). Weight at the time of measurement was also not included because of its correlation with animal age.
Temperament could be another culling criterion together with production after first lactation. To evaluate the impact of culling of females due to poor temperament on genetic parameter estimates, another dataset was constructed using only heifers and primiparous. A total of 1942 reactivity records from 1138 females were used for this analysis, and the relationship matrix contained 12823 animals. Reactivity data from first-lactating females were submitted to a univariate analysis using the same method described above.
Test-day milk production was used in a bivariate analysis. Only records from 380 females that represented the test-day recorded nearest the reactivity measurements were included. The relationship matrix comprised 12947 animals.
Around 45% of the females were in the first two categories of visual (TS) and reactivity (TR) scores for the Guzerá herds studied. The overall mean, median, and mode of the reactivity (TR) were 1248 ± 1354; 727 ± 26.7; and 270 points. It was assumed that the movements of an animal while in the weighing scale were associated with its temperament. TR mean, range and median were 1406 ± 1472 points (168 to 11242) and 866 ± 41, respectively, in the rainy season, and 1086 ± 1201 (161 to 8728) and 597 ± 34, respectively, in the dry season.
Bartlett’s test revealed no significant difference (P>0.05) between TR variances measured in the two seasons, and the data were therefore analysed together. Pearson’s correlation showed a positive coefficient of 0.71 between the temperaments measured by visual scores (TS), and reactivity values (TR). TR had a standard deviation that proportionally varied with its mean and showed no normal distribution (Skewness = 2.54 and Kurtosis = 8.22) as did TS (Skewness = 0.77 and Kurtosis = -0.49). Therefore TR was transformed to a logarithmic scale in base 10, resulting in a quasi-normal distribution and a reduced mean/variance relationship, meeting the assumption of a normal distribution for analysis of variance.
Analysis of variance for base 10-log reactivity showed significant effects (P < 0.001) for herd, season, physiological state, weight class, entry order class, and age in days (Table 3). The linear and quadratic effects of age as a covariate were also significant.
The trend in reactivity according to female age is shown in Figure 1. The reactivity decreased with age. The linear and quadratic regression coefficients for age upon log-reactivity were approximately -0.004 and 0.000005 per day, respectively. A negative linear effect indicates that reactivity first tended to decrease as the age of the females increased, whereas the significant positive quadratic term indicates a lower regression coefficient that tends to become almost constant at older ages.
|
Table 3. Variance analysis of base 10-log reactivity for fixed effects testing (type 3 error). |
||||
|
Source of variation |
Num DF* |
Den DF** |
F-value |
Pr>F*** |
|
Herd |
4 |
1,47 |
133 |
<0.0001 |
|
Season |
1 |
1,32 |
91 |
<0.0001 |
|
Physiological state |
1 |
2,32 |
30.5 |
<0.0001 |
|
Weight |
3 |
2,34 |
8.28 |
=0.0002 |
|
Entry order |
2 |
2,15 |
11.7 |
<0.0001 |
|
Age |
3 |
2,11 |
42.4 |
<0.0001 |
|
* Num DF: degrees of freedom of numerator, ** Den DF: degrees of freedom of denominator, |
||||
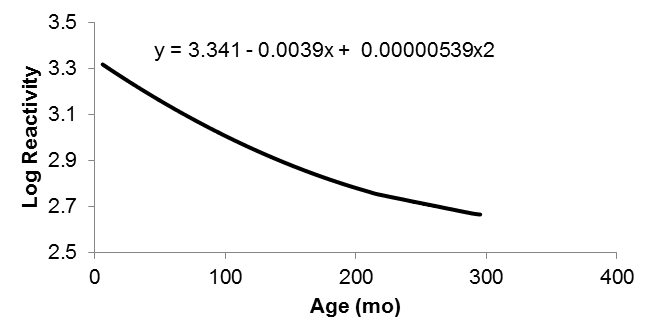 |
| Figure 1. Relationship between reactivity (base 10-Log) and age (month) of Guzerá females |
The least square means for log-reactivity across the categories for each variable are shown in Table 4. Comparisons among the categories herd, season, entry order, physiological state, weight and age revealed significant differences. Significantly higher mean reactivity values (P < 0.001) when compared with the other categories were observed for herd E, rainy season, non-lactating cows, and lower weight females. The last animals to enter the weighing scale were more reactive than the first animals. The youngest females were the most reactive.
The contrasts shown in Table 4 indicate that herds A and C were the least reactive, and they differed (P < 0.05) from the other herds. Another interesting finding was that the bulls with the most reactive daughters were not used in herd A, which showed the lowest mean reactivity. However, these bulls were used intensively in herd E, which may have contributed to the higher mean reactivity of this herd.
Likewise, mean reactivity values were similar for the first two categories of entry order and the three categories of lighter females.
|
Table 4. Least square means (LSmeans), back transformed means (in parentheses), and respective standard errors (SE) for reactivity (in points), and contrasts between LSmeans of categories for fixed effects. |
|||||
|
Fixed |
Category |
Reactivity |
SE |
T-value |
Pr>T* |
|
Herd |
A |
2.75 (563)a |
0.02 |
169 |
<0.0001 |
|
B |
2.98 (956)b |
0.03 |
108 |
<0.0001 |
|
|
C |
2.8 (646)a |
0.03 |
98 |
<0.0001 |
|
|
D |
2.9 (788)c |
0.01 |
218 |
<0.0001 |
|
|
E |
3.22 (1664)d |
0.02 |
185 |
<0.0001 |
|
|
Season |
Rainy |
2.98 (950)a |
0.01 |
241 |
<0.0001 |
|
Dry |
2.88 (768)b |
0.01 |
228 |
<0.0001 |
|
|
Entry order |
1 |
2.89 (785)a |
0.01 |
269 |
<0.0001 |
|
2 |
2.92 (833)a |
0.02 |
194 |
<0.0001 |
|
|
3 |
2.97 (954)b |
0.02 |
159 |
<0.0001 |
|
|
State |
Non-lactating |
2.97 (937)a |
0.01 |
260 |
<0.0001 |
|
Lactating |
2.89 (779)b |
0.02 |
189 |
<0.0001 |
|
|
Weight |
1 |
2.98 (950)a |
0.02 |
178 |
<0.0001 |
|
2 |
2.94 (887)ab |
0.02 |
188 |
<0.0001 |
|
|
3 |
2.92 (813)b |
0.02 |
177 |
<0.0001 |
|
|
4 |
2.87 (762)c |
0.02 |
164 |
<0.0001 |
|
|
Age |
1 |
3.07 (1217)a |
0.03 |
113 |
<0.0001 |
|
2 |
2.98 (944)b |
0.02 |
202 |
<0.0001 |
|
|
3 |
2.9 (791)c |
0.02 |
197 |
<0.0001 |
|
|
4 |
2.77 (587)d |
0.02 |
158 |
<0.0001 |
|
|
* Pr>T: probability of being higher than tabulated T-value |
|||||
The heritability and repeatability for temperament assessed by reactivity (log) were 0.06 ± 0.02 and 0.41 ± 0.03, respectively, and the visual scores (log) were 0.29 ± 0.08 and 0.4 ± 0.07 (Table 5), respectively, for the complete dataset.
|
Table 5. Variance components and genetic parameters for temperament measures and test-day milk production obtained from univariate (U) and bivariate (B) animal model analyses using Derivative Free Restricted Maximum Likelihood methodology. |
|||||||
|
Model |
Variance components and genetic parameters** |
||||||
|
σ2a |
σ2pe |
σ2e |
σ2p |
h2 (±SE***) |
R (±SE) |
rg (±SE) |
|
|
U - TR |
0.006 |
0.042 |
0.054 |
0.102 |
0.06 (±0.02) |
0.41 (±0.03) |
- |
|
U - TS |
0.006 |
0.001 |
0.014 |
0.021 |
0.29 (±0.08) |
0.4 (±0.07) |
- |
|
U - TRY |
0.006 |
0.041 |
0.059 |
0.106 |
0.06 (±0.03) |
0.39 (±0.04) |
- |
|
B - TR+TS |
0.018 (TR) |
0.031 (TR) |
0.056 (TR) |
0.105 (TR) |
0.17 (±0) |
0.3 (±0) |
0.89 (±0) |
|
0.003 (TS) |
0.005 (TS) |
0.014 (TS) |
0.022(TS) |
0.13 (±0) |
0.23 (±0) |
||
|
B - MP+TR |
1.72 (MP) |
2.48 (MP) |
2.85 (MP) |
7.05 (MP) |
0.24 (±0) |
- |
-0.46 (±0) |
|
0.002 (TR) |
0.39 (TR) |
0.014 (TR) |
0.02 (TR) |
0.08 (±0) |
- |
||
|
*TR: temperament as reactivity; TS: temperament as visual score; TRY: temperament as reactivity of heifers and first-lactating cows; |
|||||||
Genetic correlations between TR and TS revealed high value estimates (0.89). Although additive genetic variances for TR and TS using univariate (U ) analyses were equal, the variance components (σ2pe, σ2e, σ2p) were highly variable and lower for TS. The variance components obtained in the bivariate (B) analysis were different from those obtained for TS and TR in the univariate (U) analyses, with the exception of σ2e and σ2p obtained from the U-TR, U-TS and B-TR+TS models. Changes in genetic parameters were also observed whenever the bivariate model was used in the analyses. Heritability increased for TR and decreased for TS in contrast with repeatability that decreases for both traits. The results from the analysis of TR, including only data from the heifers and first-lactating females (TRY), showed no difference in the heritability (0.06 ± 0.026) or repeatability (0.39 ± 0.038) estimates. The genetic correlation between reactivity and test day milk production was estimated at -.46.
Correlation estimates suggested that TS and TR measured the same aspect of animal temperament, i.e., these traits measured the general activity of an individual. Although the electronic measuring objectively captured phenotypic variations in animal reactivity, according to Fraser (2009), it is also important to obtain subjective measurements by methods based on qualitative observations, to complement and validate the results that were objectively obtained.
The performance of females to temperament widely varied as a function of each fixed effect category. Analyses of variance were significant for all effects tested, demonstrating the complexity of reactivity. Herd was an important effect, to which implicit differences in management quality, geographic location, food availability and worker qualification can be attributed. According to Maffei et al (2006), herd was the factor that most influenced Nelore cattle reactivity, concluding that the poorer the herd management, the higher the mean reactivity, as was found in this study for herd E. The differences between this herd and others may be related mainly to some inappropriate management practices, as a consequence of both: poor infrastructure and poor stockmen training, whose importance to animal behaviour and productive performance may have been overlooked.
Positive handling experiences in early life had a strong impact on the future temperament of animals (Kabuga and Appiah 1992; Grandin, 2000; Munksgaard et al 2001; Petherick et al 2009).The impact of adopting positive handling practices was mainly observed in herds showing low reactivity (A and C) in which almost all positive handling practices were performed by caretakers who had been frequently trained and supervised (Table 1).
The best performing herds (A, C, and D) culled poor-temperament animals early from the herd. This factor may have contributed to the low prevalence of females with high mean reactivity values in those herds. In contrast, the females in herd E, in which none of these practices were implemented, showed the highest significant mean reactivity. In fact, the adoption of correct handling practices, focusing on animal welfare, appears to be one of the main ways to avoid animals with poor temperament in a herd. It should also be emphasised that cattle appreciate routines and have good memory; therefore, it is important to avoid an abusive handler from an early age (Breuer et al 2000; Munksgaard et al 2001).
Also with respect to the superiority of herd A, our data showed that bulls that produced offspring with high reactivity were not frequently used in this herd. This finding should be emphasised as it provided evidence for an association between genetic factors and the expression of reactivity as discussed by Maffei (2006) despite the low heritability observed in this study. In herd A, particularly, calves were kept with their mothers for six days after calving and kept together after milking and throughout the afternoon during lactation.
Petherick et al (2009) also suggested that unstable, inconsistent, or changing handling could have a negative effect on animal temperament. Lactating cows were brought to the milking parlour every day, which is next to the livestock scale, where they became familiar with the facilities in almost all of the farms in this study. In addition, the mother-calf relationship was partially preserved because lactating cows were kept with their calves during milking, since that, particularly in zebu dairy cattle, it is imperative to induce milk ejection (Orihuela, 1990), and human-animal contact was permitted daily, thus becoming routine, which may have also contributed to the low reactivity of the lactating cows.
With respect to the season, reactivity was found to be lowest during the dry season, despite the tolerance of Zebu cattle to higher temperatures that is usual in the summer as discussed by Hansen (2004). However, a short breeding season was adopted with some of Guzerá herds of this study to concentrate calving during the summer because of the higher forage availability during this period which, in addition to high temperatures, has high humidity. The weather conditions during the dry season, which however resemble those of the environment where the Guzerá breed evolved associated to the introduction of some specific dry season practices concerning supplementation, with more human-animal interaction, could contribute to that result.
As a consequence, females would be more reactive during the rainy season because of the high temperature-humidity index and its consequences such as the increased incidence of insects, e.g., botfly, and spittlebug that attacks Brachiaria decumbens pastures reducing forage availability and quality, as reported by Holmann and Peck (2002) in Colombia. This observation was reported also by Mandal et al (2002) for Sahiwal cattle, a Zebu breed used for dairy production. Non-lactating cows were more susceptible to stress caused by exposure to the environment and management imposed on measurement day and therefore showed higher mean reactivity values than lactating cows, as non-lactating cows were kept away from daily human contact and facilities.
To our knowledge, there are no studies that have investigated the effect of entry order on animal temperament as assessed by reactivity. This effect was significantly related to several aspects of animal behaviour, including the previous experience of the animal in the livestock containment weighing scale, changes in routine handling, and the characteristics of each animal, such as freezing behaviour.
The significance of weight class and contrasts between categories also revealed differences between the two upper classes, i.e., those classes containing low-weight females and other classes. One possibility is that heavier animals were less reactive as a consequence of a decline in agility (Petherick et al 2009).
The method of analysis used in this study identified higher levels of reactivity in the youngest animals as reported by Fordyce et al (1988) and Tözsér et al (2003). Younger animals in this experiment were maintained in paddocks with limited human contact and handling after weaning, which could contribute to the higher reactivity of this group. The animals were difficult to handle and spent more time running and making escape attempts, which was also observed by Burrow (1997) and Boivin et al (1998). However, according to Réale et al (2007), some aspects of temperament can change with age and environmental conditions, despite the fact that differences between individuals were largely maintained.
Finally, the linear and quadratic effects of age revealed the adaptation of the females to the routine handling used, which guaranteed favourable animal behaviour throughout life in the herd and satisfactory human-animal interactions as discussed by Grandin (2000). According to Boivin et al (1998), calves that had minimal human contact in early life, calves with frequent exposure to and contact with humans, and calves reared in positive handling conditions response to human handling were less fearful and easier to handle. The results highlighted the importance of decision making on the adoption of positive handling practices at early ages for obtaining animals adapted to routine handling and having an improved temperament with age.
With regards to genetic parameters, the results obtained using the complete dataset were preferred because of the slightly lower standard error of the estimates. Behavioural traits are defined in different ways, different grading scales, and different populations making it difficult to compare genetic parameters from different studies, but estimates obtained in this study generally agree with the values obtained by others (Burrow 1997; Adamczyk et al 2013).
Heritability for temperament in cattle has ranged from 0.06 to 0.53 (Burrow 1997; Adamczyk et al 2013). The low genetic parameters found in this study reinforce the importance of handling practices and environmental effects on the temperament of Guzerá females. As the additive genetic variances for TR and TS were equal, the differences in permanent environmental and residual variances were considered to be responsible for differences in the heritabilities found for each trait.
This underestimation would consequently lead to an overestimation of heritability in spite of the repeatability of this trait has remained unchanged since that parameter expresses relative contributions of genetic differences to the total phenotypic variance. In contrast, the genetic parameters obtained for TR may be biased by the intrinsic positive handling practices, adaptation to handling routines, taming of calves, and disposal or selection with a focus on temperament that reduce genetic variance in some herds; phenomenon known as phenotypic plasticity (Réale et al 2007; O’Neil et al 2010). Besides, concerns related to the data structure and size used in this study should also be considered and emphasizes the conduction of further studies with a larger sample size with less than 24-years old females to confirm the results found. With regards to genetic correlation between reactivity and test-day milk production, a moderate, negative value was found that indicated the existence of genes influencing both traits and the possibility of an indirect response to selection.
Therefore, conclusions regarding the genetic parameters, mainly TR, should be carefully drawn because of the size and design of the dataset used in this study. Considerations should also include the fact that Guzerá dairy cattle had originated from a few females chosen from Guzerá beef herds where the cows that were milked most likely were those displaying good temperament and that which contributed to the next generations. Besides, the data used in this study were taken from a sample of the whole population of Guzerá dairy cattle and may benefit in the future by a larger experimental population.
Furthermore, two of the five herds have culled the animals showing poor temperament at an early age for several years and avoided the use of bulls that produced poor-tempered offspring, which may have contributed to a decreased genetic variance. Thus, heritability for TR can also be underestimated due to positive management effects. Despite the low estimate of heritability, the extreme values found for reactivity in some paternal half-siblings could, in addition to the favourable response to the culling/selection practices used in some herds, suggest a genetic effect on this trait. Finally, the favourable, moderate genetic correlation between reactivity and test-day milk production, which would allow an indirect response to selection, should be further investigated because the joint culling/selection criteria practiced concomitantly in some herds may induce a false-negative correlation.
The authors thank Fapemig, CNPq and Pró-Reitoria de Pesquisa of UFMG for financial support, Dr. Walsiara Estanislau Maffei for providing training and equipment, and the Guzerá breeders for logistical support.
Consent for this study was obtained (Protocol number: CEUA-EGL 09/2014).
Adamczyk K, Porkoska J, Makulska J, Earley B and Mazurck M 2013 Genetic analysis and Evaluation of behavioural traits in cattle. Livestock Science 154: 1-12.
Akaike H 1974 A new look at the statistical model identification. IEEE Transactions on Automatic Control 19, 716-723.
Barrozo D, Buzanskas M E, Oliveira J A, Munari D P, Neves H H R and Queiroz S A 2012 Genetic parameters and environmental effects on temperament score and reproductive traits of Nellore cattle. Animal 6: 36-40.
Bergmann J A G, Maffei W E, Barbosa M P and Teixeira AG 2010 Measurement of animal temperament. URL, http://appft1.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&d=PG01&p=1&u=/netahtml/PTO/srchnum.html&r=1&f=G&l=50&s1=20100294204.PGNR
Boldman K G, Kriese L A, Van Vleck L D, Van Tassel C P and Kachman S D 1995 A manual for use of MTDFREML. A set of programs to obtain estimates of variances and covariances. U.S. Department of Agriculture, Agricultural Research Service, University of Nebraska, Lincoln.
Boivin X, Garel J P, Mante A and Le Neindre P 1998 Beef calves react differently to different handlers according to the test situation and their previous interactions with their caretaker. Applied Animal Behaviour Science 55: 245-257.
Bowles D, Paskin R, Gutiérrez M and Kasterine A 2005 Animal welfare and developing countries: opportunities for trade in high-welfare products from developing countries. Revue Scientifique et Technique (International Office of Epizootics) 24: 783-790.
Breuer K, Hemsworth P H, Barnett J L, Matheus L R and Coleman G J 2000 Behavioural response to humans and the productivity of commercial dairy cows. Applied Animal Behaviour Science 66: 273-288.
Burnham K and Anderson D 2003 Model Selection and Multi-Model Inference. Springer, New York, US.
Burrow H M 1997 Measurement of temperament and their relationship with performance traits of beef cattle. Animal Breeding and Abstracts 65: 478-495.
Dobson A J 2002 An Introduction to Generalized Linear Models, second ed. Chapman & Hall, London.
Fordyce G, Dodt R M and Wythes J R 1988 Cattle temperament in extensive beef herds in northern Queensland. 1. Factors affecting temperament. Animal Production Science 28: 683-687.
Fraser D 2009 Animal behaviour, animal welfare and the scientific study of affect. Applied Animal Behaviour Science 118: 108-117.
Friedrich J, Brand B and Schwerin M 2015 Genetics of cattle temperament and its impact on livestock production and breeding – a review. Archives of Animal Breeding 58: 13-21.
Goddard P J, Fawcett A R, Macdonald A J and Reid H W 2000 The behavioural, physiological and immunological responses of lambs from two rearing systems and two genotypes to exposure to humans. Applied Animal Behaviour Science 66: 305-321.
Grandin T 2000 Behavioural principles of handling cattle and other grazing animals under extensive conditions. Livestock handling and transport. CAB International Publishing, Wallingford, Oxon, UK.
Gutiérrez-Gil B, Ball N, Burton D, Haskell M, Williams J L and Wiener P 2008 Identification of quantitative trait loci affecting cattle temperament. Journal of Heredity 99: 629-638.
Hansen P J 2004 Physiological and cellular adaptations of zebu cattle to thermal stress. Animal Reproduction Science 82-83: 349-360.
Hoppe S, Brandt H R, König S, Erhardt G and Gauly M 2010 Temperament traits of beef calves measured under field conditions and their relationships to performance. Journal of Animal Science 88: 1982-1989.
Holmann F and Peck D C 2002 Economic damage caused by spittlebugs (Homoptera: Cercopidae) in Colombia: A first approximation of impact on animal production in Brachiaria decumbens pastures. Neotropical Entomology 31: 275-284.
Kabuga J D and Appiah P 1992 A note on the ease of handling and flight distance of Bos indicus, Bos taurus and their crossbreds. Animal Production 54: 309-311.
King D A, Schuehle Pfeiffer C E, Randel R D, Welsh Jr T H, Oliphint R A, Baird B E, Curley Jr K O, Vann R C, Hale D S and Savell J W 2006 Influence of animal temperament and stress responsiveness on the carcass quality and beef tenderness of feedlot cattle. Meat Science 74: 546-556.
Maffei W E, Bergmann J A G, Pinotti M, Oliveira M E C and Silva E Q 2006 Animal reactivity in a mobile cage: a new methodology to assess bovine temperament. Arquivo Brasileiro de Medicina Veterinária e Zootecnia 58, 1123-1131. Reatividade em ambiente de contenção móvel: uma nova metodologia para avaliar o temperamento bovino (in portuguese, with abstract in english).
Mandal D K, Raio A V M S, Singh K and Singh S P 2002 Comfortable macroclimatic conditions for optimum milk production in Sahiwal cows. Journal of Applied Zoology Research 13: 228-230.
Munksgaard L, DePassillé A M, Rushen J, Herskin M S and Kristensen A M 2001 Dairy cows’ fear of people: social learning, milk yield and behaviour at milking. Applied Animal Behaviour Science 73: 15-26.
O’Neil C J, Swain D L and Kadarmideen H N 2010 Evolutionary process of Bos taurus cattle in favourable versus unfavourable environments and its implications for genetic selection. Evolutionary Applications 3: 422-433.
Orihuela A 1990 Effect of calf stimulus on the milk yield of Zebu-type cattle. Applied Animal Behaviour Science 26: 187-190.
Paranhos da Costa M J R and Broom D M 2001 Consistency of side choice in the milking parlour by Holstein-Friesian cows and its relationship with their reactivity and milk yield. Applied Animal Behaviour Science 70:177-186.
Sant’Anna A C and Paranhos da Costa M J R 2013 Validity and feasibility of qualitative behavior assessment for the evaluation of Nellore cattle temperament. Livestock Science 157: 254-262.
Peixoto M G C D, Poggian C F, Verneque R S, Egito A A, Carvalho M R S, Penna V M, Bergmann J A G, Viccini L F and Machado M A 2010 Genetic basis and inbreeding in the Brazilian Guzerat (Bos indicus) subpopulation selected for milk production. Livestock Science 131: 168-174.
Petherick J C, Doogan V J, Holroyd R G, Olsson P and Venus B K 2009 Quality of handling and holding yard environment, and beef cattle temperament: 1. Relationships with flight speed and fear of humans. Applied Animal Behaviour Science 120: 18–27.
Réale D, Reader M S, Sol D, McDougall P T and Dingemanse N J 2007 Integrating animal temperament within ecology and evolution. Biological Reviews 82: 291-318.
Rueda P M, Sant’Anna A C, Valente T S and Paranhos da Costa, M P R 2015 Impact of the temperament of Nellore cows on the quality of handling and pregnancy rates in fixed-time artificial insemination. Livestock Science 177: 189-195.
Schwarz G 1978 Estimating the dimension of a model. Annals of Statistics 6, 461–464.
Schwarztzpof-Genswein K S, Shah M A, Church J S, Haley D B, Janzen K, Truong G, Atkins R P and Crowe T G 2012 A comparison of commonly used and novel electronic techniques for evaluating cattle temperament. Canadian Journal of Animal Science 92: 21-31.
Sebastian T, Watts J M, Stookey J M, Buchanan F and Waldner C 2011 Temperament in beef cattle: Methods of measurement and their relationship to production. Canadian Journal of Animal Science 91: 557-565.
Sewalen A, Miglior F and Kistemaker G J 2010 Analysis of the relationship between workability traits and functional longevity in Canadian dairy breeds. Journal of Dairy Science 93: 4359–4365.
Sutherland M A, Rogers A R and Verkerk G A 2012 The effect of temperament and responsiveness towards humans on behavior, physiology and milk production of multi-parous cows in a familiar and novel milking environment. Physiology & Behavior 107: 329-337.
Tözsér J, Maros K, Szentléleki A, Zándoki R, Nikodémusz E, Balázs F, Bailo A and Alföldi L 2003 Evaluation of temperament in cows of different age and bulls of different colour variety. Czech Journal of Animal Science 48: 344-348.
Von Keyserlingk M A G, Rushen J, Passillé A M and Weary D M 2009 Invited review: The welfare of dairy cattle-Key concepts and the role of science. Journal of Dairy Science 92: 4101-4111.
Received 16 March 2016; Accepted 19 July 2016; Published 1 September 2016