Effect of supplementation with urea or calcium nitrate and cassava leaf meal or fresh cassava leaf in an in vitro incubation using a basal substrate of sugar cane stalk
Outhen Phommasack, T R Preston* and R A Leng**
Savannakhet University, Lao PDR
Outhen@hotmail.com
* Finca Ecologica, TOSOLY, UTA (Colombia)
AA#48, Socorro, Santander, Colombia
** University of New England, NSW, Australia
Abstract
A substrate of sugar cane stalk and either cassava leaf meal or fresh cassava leaves was incubated in an in vitro system in which the source of fermentable N was calcium nitrate or urea.
Replacing urea with calcium nitrate reduced methane production on both dried or fresh cassava leaf. Increasing the fermentation time increased the production of methane, probably because of reduction of the VFA. After 5h of fermentation the methane percentage in the gas was lower with fresh than with dried cassava leaf, but there were no differences with longer fermentation times. The potential of HCN precursors in cassava leaf and roots to reduce methane production in ruminants merits further investigation.
Key words: Cassava, greenhouse gases, HCN, methane
Introduction
Methane resulting from fermentative digestion of organic matter in the rumen
represents a loss of dietary energy to the animal (Preston and Leng 2009)
and ruminants are a significant contributor to world greenhouse gas emissions (Steinfeld et al 2006). These factors have led to a global search for
strategies
to mitigate methane emissions from ruminants.
In ruminants, H2 produced in the pathways of conversion of organic matter to VFA with growth of cells, is normally removed by the reduction of CO2 to form methane. However, nitrate (present in some fresh pasture grasses) has a higher affinity for H2 than CO2 and, when it is present, H2 is effectively used in the reduction of NO3 to ammonia (NH3) thereby lowering the production of methane. The possibility of nitrate as an alternative hydrogen sink to carbon dioxide has been discouraged because of the possible toxic effects of nitrite, which is spilled into the medium under some feeding regimes (Lewis 1951).
After comprehensively reviewing the literature, Leng (2008) concluded that the inclusion of nitrate in feed supplements appeared to be entirely feasible as a means of reducing enteric methane emissions from ruminant livestock. While there is a risk of nitrite toxicity, nitrate supplementation also has potential advantages in addition to inhibiting methane emissions, viz. (i) the end product of nitrate reduction is ammonia, which for ruminants on low digestibility diets, is a major source of N for microbial growth; and (ii) nitrate reduction to ammonia is accompanied by formation of ATP from ADP and therefore theoretically microbial growth efficiency should also be improved.
The protein in cassava (Manihot esculenta, Crant) leaves is considered to be a good source of bypass protein (Ffoulkes and Preston 1978; Wanapat et al 1997, Keo Sath et al 2008). It is widely cultivated in all tropical counties and is thus a logical forage produced locally to provide the additional protein required in low protein high energy diets
Cassava contains cyanogenic glucosides, mainly linamarin (92-98%); which release hydrogen cyanide after hydrolysis in the rumen by an endogenous linamarase (Butler et al 1965; Conn 1969). Anaerobic digestion can be inhibited by cyanide, because of the high sensitivity of methanogenic bacteria to this compound (4 mg 1itre-1 in pure culture) (Eikmanns and Thauer 1984; Smith et al 1985).
Cuzin and Labat (1992) showed that additions of 5, 10, and 25 mg 1itre-l cyanide (KCN or linamarin) temporarily inhibited methanogenesis but when the concentration of cyanide reached its initial value (before KCN or linamarin addition), methane production recovered. The biodigester methanogenic microflora were sensitive to cyanide addition, but tolerated the low concentrations (6 mg 1itre-1) normally present in the fermenter.
As production of HCN from cyanogenic glucosides is reduced by drying (from 190 mg/kg DM in the fresh leaf to 20 mg/kg after sun-drying according to Bui Huy Nhu Phuc et al 1995), it is possible that methane production in an in vitro fermentation would be less when the substrate contained fresh cassava leaves rather than dried leaves.
The purpose of the present study was to determine methane production from a diet based on sugar cane stalk as substrate supplemented with protein from cassava leaf meal or fresh cassava leaf using calcium nitrate and urea as sources of non-protein nitrogen.
Hypothesis
-
Methane production in an in vitro fermentation (artificial rumen) of sugar cane stalk would be reduced when calcium nitrate replaced urea as NPN source, and fresh cassava leaf was the protein source rather than cassava leaf meal.
Materials and Methods
Location
The experiment was conducted in the laboratory of An Giang University, Vietnam from August to October 2010.
Treatments and experimental design
A 2 x 2 factorial design was used to evaluate the effect of (i) supplementation of cassava leaf meal or fresh cassava leaf; and (ii) supplementation of urea or calcium nitrate using sugar cane stalk as basal substrate. There were four replications of the following treatments.
- Cassava leaf meal plus urea (CLM-U)
- Cassava leaf meal plus calcium nitrate (CLM -CaN)
- Fresh cassava leaf plus urea (FCL-U)
- Fresh cassava leaf plus calcium nitrate (FCL- CaN)
Preparation of substrate and the in vitro system
The sugar cane stalk and cassava leaf meal (or fresh cassava leaf) were milled in a coffee grinder and mixed with the sources of N (urea or calcium nitrate). Recycled water bottles (capacity 1500 ml) were used for the incubation and collection of the gas (Photo 1). A hole was made in the lid of each of the bottles, which were inter-connected with a plastic tube (id 4mm). The bottle receiving the gas had the bottom removed and was suspended in a larger bottle (5 litre capacity) partially filled with water, so as to collect the gas by water displacement. The bottle that was suspended in water was calibrated at 50 ml intervals to indicate the volume of gas. A representative sample (12 g DM) of the mixtures (Table 1) was put in the incubation bottle, to which was added 0.96 litres of buffer solution (Table 2) and 240ml of rumen fluid from a buffalo. The rumen fluid was taken at 10-11pm in the slaughter-house from a buffalo immediately after the animal was killed. A representative sample of the rumen contents (including feed residues) was put in a vacuum flask and stored until 8-9am the following morning when the contents were filtered through one layer of cloth before being added to the incubation bottle. The remaining air in the flask was flushed out with carbon dioxide. The incubation flask was connected by a plastic tube (id 3mm) to a second flask (a calibrated recycled water bottle with the bottom removed) suspended in water so as to measure the gas production by water displacement. The incubation flasks were then incubated at 38°C for 43h.
|
Table 1. Ingredients (g) in the substrate |
||||
|
CLM-U |
FCL-U |
CLM-CaN |
FCL-CaN |
|
|
Sugar cane stalk |
8.76 |
8.76 |
8.54 |
8.54 |
|
Cassava leaf meal |
3.00 |
3.00 |
|
|
|
Fresh cassava leaf |
3.00 |
|
3.00 |
|
|
Urea |
0.24 |
0.24 |
|
|
|
Ca(NO3)2.4H2O |
0.46 |
0.46 |
||
|
Table 2. Ingredients of the buffer solution (adapted from Tilly and Terry 1964) |
|||||||
|
Ingredients |
CaCl2 |
NaHPO4.12H2O |
NaCl |
KCl |
MgSO4.7H2O |
NaHCO3 |
Cysteine |
|
(g/liter) |
0.04 |
9.30 |
0.47 |
0.57 |
0.12 |
9.80 |
0.25 |
|
|
|
|
Photo 1.
The in vitro fermentation
system using recycled water |
Photo 2.
Measurement of
percentage |
Data collection and measurements
Gas production was measured after 5, 15 and 43h and samples analyzed for methane percentage using an infra-red analyser (Crowcon Instruments Ltd, UK) (Photo 2). Each time, after the gas volume was measured and analyzed for methane, the gas was released to empty the bottles for the next measurement. Residual undigested substrate in the incubation bottle was determined by filtration through cloth and drying of the residue in the oven at 105°C for 24h.
Statistical analysis
The data were analyzed by
the General Linear Model (GLM) option in the ANOVA program of the Minitab
Software (version13.2) (Minitab 2000). Sources of variation in the model
were: N source, cassava leaf, interaction N*cassava leaf and error.
Results and Discussion
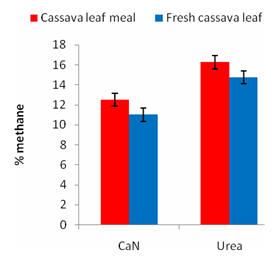
Gas production was higher for fresh than for dried cassava at 5h and for urea versus nitrate at 15h; however, there were no treatment differences at other sampling times and only a tendency (P=0.15) for total gas production at 43h to be higher on urea than on nitrate (Table 3). After 5h (Figure 1) and at all subsequent sampling times the methane production was lowered in presence of nitrate compared with urea. Comparing fresh with dried cassava leaves, after 5h the methane percentageproduction was lower for the former both before and after correcting the data for differences in gas volume (Figure 2). However, there were no differences at 15 and 43 h, and no differences at all times for volume of methane produced. The proportion of substrate fermented, and the gas production per unit substrate fermented, were not affected by the treatments. The production of methane as total volume per unit substrate fermented (Figure 3), was lower for nitrate than for urea. There were no differences between fresh and dried cassava leaf (Figure 4). There were no interactions between N source and cassava leaf processing for any of the measurements.
|
Table 3. Mean values for gas production and production of methane after 5, 15 and 43h of in vitro incubation of fresh sugarcane in presence of urea or calcium nitrate and fresh or dried cassava leaves |
|||||||
|
|
Effect of cassava |
Effect of N source |
P values |
SEM |
|||
|
|
CLM |
FCL |
CaN |
Urea |
Cassava |
N source |
|
|
0- 5h |
|
|
|
|
|
|
|
|
Gas production, ml |
875 |
975 |
950 |
900 |
0.030 |
0.24 |
28.9 |
|
Methane, % |
14.4 |
12.9 |
11.8 |
15.5 |
0.040 |
<0.001 |
0.46 |
|
Methane, ml |
125 |
125 |
111 |
138 |
0.92 |
<0.001 |
4.46 |
|
5-15h |
|
|
|
|
|
|
|
|
Gas production, ml |
925 |
900 |
831 |
994 |
0.71 |
0.03 |
47.2 |
|
Methane, % |
19.6 |
19.3 |
16.5 |
22.4 |
0.68 |
<0.001 |
0.62 |
|
Methane, ml |
182 |
174 |
136 |
220 |
0.35 |
<0.001 |
5.99 |
|
15-43h |
|
|
|
|
|
|
|
|
Gas production, ml |
450 |
444 |
425 |
469 |
0.88 |
0.30 |
28.9 |
|
Methane, % |
37.4 |
37.0 |
34.9 |
39.5 |
0.74 |
<0.001 |
0.79 |
|
Methane, ml |
167 |
164 |
147 |
184 |
0.81 |
0.01 |
8.41 |
|
At 43h |
|
|
|
|
|
|
|
|
Total gas production, ml |
2250 |
2319 |
2206 |
2363 |
0.51 |
0.15 |
72.3 |
|
Total methane, ml |
474 |
464 |
395 |
543 |
0.04 |
<0.001 |
13.9 |
|
Overall methane, % |
21.0 |
19.9 |
17.9 |
23.0 |
0.60 |
<0.001 |
0.33 |
|
DM substrate fermented, g |
6.56 |
6.89 |
6.67 |
6.78 |
0.21 |
0.68 |
0.18 |
|
DM substrate fermented, % |
52.4 |
57.2 |
54.8 |
54.9 |
0.19 |
0.97 |
2.39 |
|
Gas production, ml/g substrate DM fermented |
343 |
337 |
331 |
348 |
0.79 |
0.56 |
16.9 |
|
Methane, ml/g DM substrate fermented |
72.6 |
67.6 |
59.8 |
80.4 |
0.27 |
<0.001 |
3.08 |
|
|
|||||||
|
|
|
|
Figure 1.
Effect of CaN vs urea on methane content of gas |
Figure 2.
Effect of fresh vs dry cassava leaf on methane content of |
 |
 |
|
Figure 3.
Effect of CaN vs urea on methane per unit substrate fermented after 43h incubation |
Figure 4.
Effect of fresh cassava leaf or dried cassava leaf meal on methane per unit substrate fermented after 43h incubation |
The lowering in methane production when nitrate, compared with urea, was the NPN source with an energy substrate of sugar cane stalk, mirrored the findings of Sangkhom et al (2011) who used cassava root as the substrate. Comparable comparisons for cassava root versus sugar cane showed the proportion of substrate fermented to be higher, while the production of methane per unit substrate fermented was lower, for the former.
The values recorded in this study for total production of methane during the incubation, and amount of methane produced per unit DM substrate fermented, were higher than those reported by Le Thi Ngoc Huyen et al (2010). The differences can be attributed to the nature of the substrate: NaOH-treated rice straw in the experiment of Le Thi Ngoc Huyen et al (2010) compared with the more digestible sugar cane stalk in the present study.
The linear increase in the methane content of the gas with fermentation time (Figures 5 and 6) showed the same tendency as was reported by Sangkhom et al (2011). As discussed by those authors, these changes almost certainly reflected changes in the nature of the fermentation as carbohydrate was fermented first to VFA, methane and carbon dioxide followed by secondary fermentation of the VFA to methane and carbon dioxide.