Highly pathogenic avian influenza risk, biosecurity and smallholder adversity
J Otte, D Pfeiffer*, T Tiensin**, L Price*** and E Silbergeld***
FAO, Via delle terme di Caracalla, 00153 Rome,
Italy
joachim.otte@fao.org
* Royal Veterinary College, Hatfield, Herts, AL9
7TA, UK
pfeiffer@rvc.ac.uk
** Utrecht University, Yalelaan 7, 3584 CL Utrecht,
NL
t.tiensin@vet.uu.nl
*** Johns Hopkins University Bloomberg School of
Public Health, 615 N. Wolfe Street, E6644 Baltimore, MD 21205,
USA
lprice@jhsph.edu and esilberg@jhsph.edu
Abstract
There is considerable global concern over the newly emergent H5N1 strain of avian influenza that has affected millions of domestic poultry flocks and resulted in more than 150 deaths in humans. There has been little analysis of the general assumption that smallholder backyard poultry flocks are inherently at higher risk of highly pathogenic avian influenza (HPAI) than confined and commercial scale operations. We utilized data from Thailand, collected in 2004, to test the relative risks of HPAI infection in poultry flocks, by species, type of operation, and geographic location. The results indicate that backyard flocks are at lower risk of HPAI infection compared to commercial scale operations of broiler or layer chickens or quail. These findings are plausible in terms of the opportunities for breach of biosecurity in commercial scale, industrial operations.
Both experimental and observational studies in developed country settings have demonstrated the capacity of microbes to enter and leave these larger operations despite the implementation of standard biosecurity measures. Patterns of infection during 2002 Newcastle disease epidemic in Denmark proved further evidence to question that smallholder backyard flocks are at higher risk of epidemic diseases than commercial operations. These results should be considered by policy makers and public health officials when developing plans to control or prevent HPAI in order to limit avoidable adverse effects on the livelihood of smallholder poultry producers in developing countries.
Key words: Backyard, biosecurity, commercial poultry, highly pathogenic avian influenza (HPAI), policy, poultry, public health, risk, smallholders
Introduction
Outbreaks of highly pathogenic avian influenza (HPAI) were first reported in Southeast Asia in late 2003, although the current emergence of the H5N1 virus is now considered to have occurred as early as 1996 when it was first identified in geese in Guangdong province in southern China (Sims et al 2005). Since then it has spread rapidly and over large distances, with outbreaks occurring in domesticated poultry and some wild bird populations in Mongolia, southern Russia, the Middle East and, in 2005, in Europe and Africa. To date, several epidemic waves have occurred in Indonesia, Thailand, Vietnam, and Laos. The widespread practice of smallholder backyard poultry keeping in many of these countries is frequently cited as one of the primary risk factors for these outbreaks, including infections in humans, and the persistence of the virus in domestic poultry populations. Thus, some governments are, amongst other measures, considering the prohibition of unconfined poultry flocks in order to increase 'biosecurity' of smallholder backyard production. Many of these measures may be prohibitively expensive for resource-poor smallholder producers and thereby could force them to abandon poultry keeping. Given these likely adverse impacts of restrictive policies on smallholder poultry growers, it is important to examine the evidence base for such measures in terms of their effect on risks of HPAI.
Very few of the measures proposed to enhance
biosecurity of poultry operations have been rigorously tested for
their effectiveness against HPAI. There is an assumption that
because the majority of HPAI outbreaks have been reported in
smallholder backyard flocks, these operations are inherently more
risky than other types of poultry operations. In order to
critically examine this assumption we utilize available data from
the HPAI epidemic in Thailand, review recent studies on pathogen
movement in and out of standard commercial poultry facilities in
the USA and Western Europe, and draw on findings of the 2002
Newcastle disease (ND) epidemic in Denmark.
The relative risk of HPAI in commercial vs backyard flocks in Thailand
In late 2003, poultry farms in some regions of Thailand experienced massive mortality and in mid-January 2004 the Thai animal health authorities launched a nationwide active surveillance programme to detect possible HPAI infections in poultry based on laboratory testing of cloacal swabs, an exercise that was repeated in October 2004, referred to as X-ray survey. In the course of these active surveillance programmes, swabs were collected from around 230,000 flocks (5 birds per flock) from more than 50,000 villages (4 flocks per village) and tested for avian influenza virus. Furthermore 72,000 serum samples were collected for diagnosis.
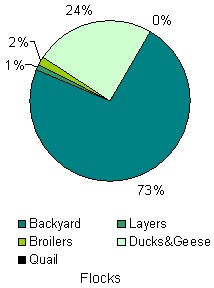
In order to examine whether backyard poultry operations are at higher risk of HPAI infection than 'commercial' flocks we draw on published data on the 2004 HPAI epidemic in Thailand (Tiensin et al 2005) and compare the incidence of HPAI infection in 'backyard flocks' with that in flocks classified as 'non-backyard / commercial' by the Thai Department for Livestock Development (DLD). In the case of chicken the latter were further classified as 'layer' or 'broiler' flocks, while for other poultry, flocks were classified by species, namely as 'duck', 'geese' or 'quail' flocks. Given the similar methods of production and nature of ducks and geese we aggregate the latter into one group in the following figures. As shown in Figures 1 and 2, the Thai poultry industry is complex in terms of bird species and production.
|
|
|
|
Figure 1. Contribution of different 'flock
types' to total domestic |
Figure 2. Contribution of different 'flock
types' to total number |
Commercial enterprises of broiler or layer chickens comprise most of the standing poultry population in Thailand while backyard flocks constitute most of the flocks. Backyard flocks, which consist of 30 birds per flock on average, constitute approximately three quarters of flocks but account for only around one fifth of the standing poultry population. Commercial broiler enterprises (having 3,500 birds per flock on average) constitute only two percent of all 'flocks' but account for nearly 60 percent of the standing poultry population. In the case of quail, although these may be grown in backyard operations, the majority are reared in confinement as larger flocks, primarily for egg and subsequently meat production. Average size of quail flocks is around 1,400 birds, confirming the large proportion of commercial operations.
The Thai poultry industry is also not uniformly distributed throughout the country, with large-scale commercial production being particularly important in the central and eastern region. The average size of duck and geese flocks is below 50 and 5 birds respectively in the North, Northeast and South, indicating that they are mainly backyard operations, while in the central and eastern region average flock size is 240 and 340 respectively for ducks and 80 and 110 for geese, suggesting that in these regions they are to a large extent commercial operations. The majority of quail also appear to be commercially raised, average flock size ranging from 170 birds in the southern region to 9,000 birds in the central region.
A total of 1,769 flocks with HPAI infection were reported to / detected by the Thai animal health authorities in 2004 (Tiensin et al 2005, do not distinguish infection detected by the active surveillance programmes from infection detected by disease reporting). The distribution of these infections by flock type is shown in Figure 3, which shows that over 50 percent of the registered infections involved backyard flocks. However, the proportional contribution of different flock types to registered infections (number of flocks affected by HPAI) is markedly different from their contribution to the total number of flocks. The crude risk of infection, expressed as a percentage of the flock type, is shown in Figure 4.
|
|
|
|
Figure 3. HPAI infections registered in
Thailand in 2004 |
Figure 4. Risk of infection (%) with HPAI in
Thailand in 2004 |
Quail flocks show the highest risk of detected infection, nearly reaching 1.6 percent, followed by layer and broiler flocks, both with infection risks of just above 0.2 percent. Against expectations, backyard flocks show the lowest risk of detected infection with HPAI (0.05 percent), only one quarter that of layer and broiler flocks. These results may reflect differences in ascertainment. It is likely that HPAI is more readily detectable by the personnel in large commercial operations and more likely to be brought to the attention of animal health authorities. However, given the attention the DLD paid to backyard operations, and the active surveillance programmes that were in place in Thailand in 2004, this potential ascertainment bias is unlikely to be the sole explanation for the higher risk of HPAI infection being detected in layer and broiler flocks compared to backyard operations.
Another explanation for these differences may be due to other risk factors related to HPAI, such as ecological and landscape factors which might modulate transfers from wild avians and among domesticated flocks. From a temporal and a geographical perspective, HPAI outbreaks did not occur uniformly or randomly across Thailand in 2004, but were shown to be linked to certain agro-ecological factors such as the extent of wetlands and rice paddies (Gilbert et al 2006). The central region was particularly affected, followed by the eastern region, while the northern, northeastern and southern regions only experienced minor epidemics. Given that these different regions also have different mixtures of flock types, the data was subjected to statistical multivariate analysis to control for potential confounding and describe potential interactions between region and flock type specific risks within region and species category. Table 1 displays the adjusted odds ratios (and their 95% confidence intervals) for the selected risk factors, taking backyard operations in the northeastern region (lowest crude risk) as the reference group (odds ratio = 1).
|
Table 1. Odds ratios and 95% confidence intervals for selected risk factors for HPAI infection in Thailand in 2004 |
|||
|
Region |
Flock type |
OR |
95%CI |
|
Northeast |
Backyard |
1.0 |
|
|
Layers |
5.0 |
2.3-11.5 |
|
|
Broilers |
1.1 |
0.3-4.4 |
|
|
Ducks |
0.3 |
0.2-0.6 |
|
|
Quail |
57.0 |
7.9-410.8 |
|
|
Geese |
1.6 |
0.2-11.2 |
|
|
North |
Backyard |
10.8 |
8.7-13.5 |
|
Layers |
83.1 |
54.7-126.1 |
|
|
Broilers |
115.9 |
80.9-166.0 |
|
|
Ducks |
17.4 |
13.0-23.3 |
|
|
Quail |
1064.1 |
570.1-1986.1 |
|
|
Geese |
29.1 |
10.7-79.2 |
|
|
Central |
Backyard |
27.1 |
19.6-31.1 |
|
Layers |
79.1 |
55.0-113.9 |
|
|
Broilers |
104.5 |
74.7-146.1 |
|
|
Ducks |
127.8 |
101.8-160.5 |
|
|
Quail |
1044.4 |
666.7-1636.2 |
|
|
Geese |
54.4 |
25.0-106.0 |
|
|
East |
Backyard |
19.5 |
11.9-20.7 |
|
Layers |
42.7 |
24.3-74.9 |
|
|
Broilers |
14.7 |
7.2-30.3 |
|
|
Ducks |
19.4 |
12.8-29.4 |
|
|
Quail |
- |
|
|
|
Geese |
39.0 |
12.3-123.4 |
|
|
South |
Backyard |
2.1 |
1.02-2.3 |
|
Layers |
1.6 |
0.2-11.2 |
|
|
Broilers |
5.8 |
1.8-18.4 |
|
|
Ducks |
1.1 |
0.6-2.2 |
|
|
Quail |
6.7 |
0.9-47.8 |
|
|
Geese |
- |
|
|
The multivariate statistical analysis shows that there was an interaction between region and species, such that within the North and Central regions, amongst the flock types in the region, backyard flocks had the lowest odds ratios of outbreak occurrence. In the East, there was relatively little difference between flock types, but layers and geese had the highest odds ratios. In the South, the odds ratios of infection were not different from backyard flocks in the Northeast for layers, ducks and quail, but they were higher for backyard and broiler flocks. In the Northeast, layers and quail had higher odd ratios than backyard flocks, whereas they were reduced in ducks, layers, broilers, ducks and geese in the South and Northeast. Across regions, the odds ratios for quail flocks to experience HPAI infection are by far the highest. These results reflect the geographical pattern of the outbreaks most of which occurred in the central region of the country, and the lowest number in the South.
Taken together, the data do not support the
assumption that backyard poultry production in Thailand is more
risky, in terms of HPAI infection, than larger and confined
commercial poultry operations (that is, quail, or either layer, or
broiler chickens). There are no data that permit examination of
production factors, apart from flock type and species, which might
also contribute to increasing or decreasing HPAI infection risks
within and between flock types. It is therefore prudent to
re-examine the assumption that significant reductions in HPAI risk
will be achieved by imposing operational changes, such as
confinement on smallholder backyard poultry producers. Moreover,
the assumption that the standard operations of large(r)-scale
commercial poultry producers are highly bio-secure also needs
re-examination.
The limits of biosecurity
Biosecurity has to be viewed in the context of production systems as well as practices in poultry production. Even though backyard flocks are not subject to specific measures to enhance biosecurity, they may nevertheless be less at risk for some infections than commercial operations. In fact, large-scale poultry operations pose significant challenges to ensuring biosecurity stemming from the confinement of large numbers of birds (as many as 50,000 in modern broiler houses), which imposes the need to supply these large populations kept at high densities with feed, water and air as well as the need to dispose of their wastes. It is important to note that because confinement of thousands of animals requires controls to reduce heat and regulate humidity, poultry and swine houses require high volume ventilation which results in considerable movement of materials from and into the external environment (Jones et al 2005). The other major challenge to biosecurity arises through the need to dispose of large amounts of animal waste from these large populations - each broiler chicken is estimated to produce about 1.7 kg waste over its 6-7 week lifespan.
The challenge to biosecurity on poultry farms can be discussed using two poultry diseases of global significance, campylobacteriosis and ND, as examples. Inferences are relevant to understand the opportunities for pathogen transfers in and out of confined poultry operations. ND is transmitted among poultry via contaminated faeces and probably via inhalation of aerosols, which is similar to HPAI. The specific mechanisms for spread between farms are also similar to HPAI, i.e. movement of poultry, poultry products, humans, contaminated feed and water. A major difference is the availability of vaccines for ND which can effectively control the disease.
Studies of Campylobacter, an avian commensal and human pathogenic bacteria, are informative as well. Like avian influenzas, wild birds are the natural vertebrate reservoirs of Campylobacter spp, and can serve as vectors for transmission to other vertebrates (eg. Cabrita et al 1992; Fernandez et al 1996; Yogasundram et al 1989). Campylobacter spp move among avian host species, both domesticated (van den Bogaard and Stobberingh 2000) and wild (Broman et al 2004; Petersen et al 2001). The exchange of Campylobacter between broiler flocks and wild avians can occur in both directions (Craven et al 2000). In confinement, broiler poultry are readily colonized by Campylobacter, and the external environment appears to be a major source of colonization. The importance of the environmental reservoir and the inability of conventional biosecurity measures to prevent the movement of microbes in and out of modern broiler facilities were both demonstrated in a recent study of Campylobacter-free broiler flocks, housed in sanitized facilities, using standard biosecurity measures, and fed Campylobacter-free feed and water. Seven out of ten of these flocks became colonized with Campylobacter by the time of slaughter and two flocks were colonized by Campylobacter strains genetically indistinguishable from strains isolated from puddles outside of the facility prior to flock placement (Bull et al 2006). Although the route of entry was not determined, this study clearly showed the capacity for microbes to enter broiler facilities despite the implementation of standard biosecurity measures.
Once a poultry flock is colonized with Campylobacter, the food, water and air within the house quickly becomes contaminated with the bacterium (ibid). Contaminated air exiting the house via ventilation systems is a potential source of Campylobacter to the external environment, and microbes may be carried some distances by wind and surface water transport. Campylobacter strains with identical DNA fingerprints to those colonizing broilers have been measured in air up to 30 m downwind of broiler facilities housing colonized flocks (ibid). There are additional mechanisms by which Campylobacter and other microbes enter and leave 'bio-secure' poultry houses. For example, insects may carry microbes in and out of facilities through ventilation systems and small openings. This was demonstrated in a study in Denmark which found that Campylobacter carriage was common among flies surrounding the broiler facilities and that as many as 30,000 flies may enter a broiler facility during a single flock rotation in the summer months (Hald et al2004). House flies captured within broiler facilities and other food environments can also carry multi-drug resistant bacteria (Macovei and Zurek 2006) as well as avian influenza virus (Bean et al 1985).
Animal house wastes constitute another pathway for
pathogens to exit poultry houses. In large scale operations, with
few exceptions poultry wastes are managed by land disposal. Some
pathogens, including viruses, can survive in poultry wastes for
considerable amounts of time (Gerba and Smith 2005). Land-disposed
poultry house wastes are attractive to wild birds due to the
presence of spilled feed in these wastes. These then may become
infected and contaminate water supplies of other poultry
operations. In addition, poultry house wastes are used in
aquaculture as 'bedding' in many countries around the world. This
practice provides an opportunity for direct contact by wild
waterfowl. The shipment of poultry wastes for this purpose has been
suggested by some as a possible mechanism for the transfer of HPAI
from Asia to Central Europe (Butler 2006).
Newcastle disease in Denmark
The 2002 ND epidemic, which has been very well researched and documented by the Danish Veterinary and Food Administration (2003), provides valuable information on the relative risk of introducing epidemic disease agents into commercial vs. backyard poultry operations. Similar to Thailand, the vast majority (app 90%) of poultry in Denmark is raised by commercial enterprises while smallholder backyard poultry keepers constitute the great majority of poultry producers (>95%) (Mortensen, personal communication).
Several incursions of ND have been recorded in
Denmark over the past years, which, however, only led to a major
epidemic in 2002. Between July and August of 2002, at total of 135
outbreaks (four primary outbreaks and 131 secondary outbreaks) of
ND were detected, of which all but one occurred in the central and
southern part of Jutland. The majority of outbreaks (126) were
observed in backyard flocks while only nine commercial flocks
(layers or pullets) were infected. However, the areas affected by
the epidemic only contained 270 registered commercial flocks
against nearly 23,000 backyard flocks. Thus, the risk of infection
was around 50 times higher for commercial than for backyard flocks.
Moreover, tracing of outbreaks revealed that all infections in
backyard flocks originated from one of the four primary outbreaks
(all in commercial flocks), via movement of live birds. The source
of infection of the primary outbreaks could not be determined.
However, a common feature of these four farms was that they were
all located less than 2 km from the coastline, which could be a
potential risk factor for infection from wild waterfowl. In one
case, a possible path of entry for the ND virus was a ventilator,
which had been put on suction instead of exhaustion after repair,
and in another case the virus may have been introduced through feed
from a feed silo, which had not been covered.
Conclusions
-
Biosecurity is defined as any practice or system that prevents the spread of infectious agents from infected to susceptible animals, or prevents the introduction of infected animals into a herd, region, or country in which the infection has not yet occurred (Radostits 2001) Another, more strict definition has been proposed, which states that biosecurity is the outcome of all activities undertaken by an entity to preclude the introduction of disease agents into an area that one is trying to protect (Dargatz et al 2002). Although a combination of measures may significantly reduce the risk of pathogen introduction and spread, for a variety of pathogens 'zero' risk is virtually impossible to achieve in farmed livestock populations, even in commercial farms in highly developed settings.
-
Practical implementation of biosecurity requires measures to be tailored to the pathogen(s) which constitute(s) the threat as well as to the production practices of the farming system at risk. This requires identification of the main pathways of pathogen transmission, quantification of risks and assessment of efficacy and cost of proposed risk mitigation measures. The imposition of measures which do not significantly reduce the risk of pathogen introduction but place severe economic burdens on society or groups thereof may be politically opportune but are unjustifiable, as they do not reduce public health risk.
-
The Thai data suggests that production methods utilized in many non-backyard / commercial poultry operations, with less than perfect biosecurity, apparently increase risks of HPAI infection, at the farm or flock level, above those experienced by subsistence backyard producers. The risk of infection for an individual farm is the combined result of its risk mitigation and its risk 'propagation' measures. Isolation of birds, e.g. through housing is an example of the former, 'traffic' onto the farm, e.g. related to the purchase of feed, is an example of the latter. On average, smallholder poultry operations may therefore be a lower risk of disease introduction than larger commercial operations, as in the latter, the risk mitigating measures may not outweigh the risk propagating practices. Thus, although the majority of reported HPAI outbreaks in Thailand in 2004 occurred in the latter, this increased cumulative risk of HPAI in the smallholder sector is primarily due to their relatively greater numbers rather than more risky production practices.
-
Some of the measures being considered to make subsistence poultry production 'safer', e.g. forced housing or confinement of poultry, as practiced by large scale commercial producers, are not likely to result in a major reduction of HPAI risks. In contrast, the costs will be very high and will be imposed upon a marginal group of entrepreneurs and household producers. This may lead to an overall reduction of HPAI outbreaks, but more as a result of the loss of household production flocks than as a result of enhanced biosecurity.
References
Bean W, Kawaoka Y, Wood J, Pearson J and Webster R 1985 Characterization of virulent and avirulent A/chicken/Pennsylvania/83 influenza A viruses: Potential role of defective interfering RNAs in nature. Journal of Virology 54: 151-160. http://jvi.asm.org/cgi/reprint/54/1/151
Broman T, Waldenstrom J, Dahlgren D, Carlsson I, Eliasson I and Olsen B 2004 Diversities and similarities in PFGE profiles of Campylobacter jejuni isolated from migrating birds and humans. Journal of Applied Microbiology 96: 834-843.
Bull S A, Allen V M, Domingue G, Jorgensen F, Frost J A, Ure R, Whyte R, Tinker D, Corry J E L, Gillard-King J and Humphrey T J 2006 Sources of Campylobacter spp. colonizing housed broiler flocks during rearing. Applied and Environmental Microbiology 72: 645-652. http://aem.asm.org/cgi/reprint/72/1/645.pdf
Butler D 2006 Doubts hang over source of bird flu spread. Nature 439(7078): 772.
Cabrita J, Rodrigues J, Braganca F, Morgado C, Pires I and Goncalves A 1992 Prevalence, biotypes, plasmid profile and antimicrobial resistance of Campylobacter isolated from wild and domestic animals from northeast Portugal. Journal of Applied Bacteriology 73: 279-285.
Craven S, Stern N, Line E, Bailey J, Cox N and Fedorka-Cray P 2000 Determination of the incidence of Samonella spp., Campylobacter jejuni and Clostridium perfringens in wild birds near broiler chicken houses by sampling intestinal droppings. Avian Diseases 44: 715-720.
Danish Veterinary and Food Administration 2003 Newcastle disease outbreaks in Denmark 2002 - Final report from www.foedevaredirektoratet.dk
Dargatz D A, Garry F B and Traub-Dargatz J L 2002 An introduction to biosecurity of cattle operations. Veterinary Clinics of North America Food Animal Practice 18: 1-5.
Fernandez H, Gesche W, Montefusco A and Schlatter R 1996 Wild birds as reservoir of thermophilic enteropathogenic Campylobacter species in southern Chile. Memorias Instituto Oswaldo Cruz 91: 699-700.
Gerba C and Smith J E 2005 Sources of pathogenic microorganisms and their fate during land application of wastes. Journal of Environmental Quality 34: 42-48. http://jeq.scijournals.org/cgi/reprint/34/1/42.pdf
Gilbert M, Chaitaweesub P, Parakamawongsa T, Premashthira S, Tiensin T, Kalpravidh W, Wagner H and Slingenbergh 2006 Free-grazing ducks and highly pathogenic avian influenza, Thailand. Emerging Infectious Diseases 12: 227-234.
Hald B, Skovgard H, Dang Duong Bang, Pedersen K, Dybdahl J, Jespersen J B and Madsen M 2004 Flies and Campylobacter infection of broiler flocks. Emerging Infectious Diseases 10: 1490-1492 http://www.cdc.gov/ncidod/eid/vol10no8/pdfs/04-0129.pdf
Jones T, Donnelly C and Stamp Dawkins M 2005 Environmental and management factors affecting the welfare of chickens on commercial farms in the United Kingdom and Denmark stocked at five densities. Poultry Science 84: 1155-1165.
Macovei L and Zurek L 2006 Ecology of antibiotic resistance genes: characterization of enterococci from houseflies collected in food settings. Archives of Environmental Microbiology 72: 4028-4035.
Petersen L, Nielsen E M, Engberg J, On S L W and Dietz H H 2001 Comparison of Genotypes and Serotypes of Campylobacter jejuni Isolated from Danish Wild Mammals and Birds and from Broiler Flocks and Humans. Applied and Environmental Microbiology 67: 3115-3121 http://aem.asm.org/cgi/reprint/67/7/3115
Radostits O M 2001 Control of infectious diseases of food-producing animals. In: Herd Health: Food animal production medicine, 3rd Edition, Saunders Company.
Sims L D, Domenech J, Benigno C, Kahn S, Kamata A, Lubroth J, Martin V and Roeder P 2005 Origin and evolution of highly pathogenic H5N1 avian influenza in Asia. Veterinary Record 157: 159-164.
Tiensin T, Chaitaweesub P, Songserm T, Chaising A, Hoonsuwan W, Buranathai C, Parakamawongsa T, Premashthira S, Amonsin A, Gilbert M, Nielen M and Stegeman A 2005 Highly pathogenic avian influenza H5N1, Thailand 2004. Emerging Infectious Diseases 11: 1664-1672.
van den Bogaard A E and Stobberingh E E 2000 Epidemiology of resistance to antibiotics. Links between animals and humans. International Journal of Antimicrobial Agents 14: 327-335.
Yogasundram K, Shane S and Harrington K 1989 Prevalence of Campylobacter jejuni in selected domestic and wild birds in Louisiana. Avian Diseases 33: 664-667.
Received 28 April 2007; Accepted 5 June 2007; Published 6 July 2007