Physical and chemical quality characteristics of warthog (Phacochoerus africanus) meat
L C Hoffman and J Sales
Department of Animal Sciences, University of Stellenbosch, PO Bag X1, Matieland, 7602, South Africa
lch@sun.ac.za
Abstract
The warthog (Phacochoerus
africanus), a game species from Africa, has been evaluated in this study
as a potential meat source according to carcass and meat quality
characteristics.
Dressing percentage, calculated on the basis of cold carcass
weight without skin and head, presents a value of 52 %. The contribution of the
shoulder (37 %), loin (7 %), back (9 %), belly (14 %) and hind legs (32 %) to
the cold carcass weight also differ substantially from that found for the
domestic pig. However, pH60min (6.32), pH24h,
(5.49), drip loss (2.76 %), cooking loss (16.79 %), shear force (3.36 kg),
colour characteristics CIEL (46.37), CIEa* (5.06) and CIEb*
(9.21), moisture (74.04 %), lipid (1.69 %), protein (22.14 %) and ash (1.29 %)
compared well to literature values for meat from other pig species. The
fatty acid profile of the meat differs substantially from other pig species,
with unsaturated, mono-unsaturated, and polyunsaturated
fatty acid contents of 35.75, 16.70 and 47.56 %, respectively, resulting in
a polyunsaturated:saturated fatty acid ratio of 1.33.
It has
been concluded from this study that warthogs provide a meat source suitable for
human consumption that can also be promoted on its health properties.
Key words: carcass characteristics, chemical composition, physical characteristics, warthog meat
Introduction
The
warthog, (Phacochoerus africanus), belonging to the Suidae family, has
a natural distribution in Africa from Senegal and Guinea in West Africa, in the
Sudan, Sahelian zones and Guinea savannas, to Ethiopia and south to the northern
parts of the Republic of South Africa (Skinner and Smithers 1990).
Characterised by a high fecundity, the warthog has a mean of four to five
piglets per litter and a gestation period of 167 to 175 days. Mature males
can attain a body weight of 100 kg and females up to 70 kg. The growth of
warthogs (Mason 1985; Somers and Penzhorn 1992) has been documented, and Somers
(1997) calculated a sustainable harvesting rate using simulation modelling.
However, all the research conducted to date has concentrated on the biological
and conservation status of this species. Regarding meat quality
characteristics, a preliminary study (Hoffman 2001)
has found that warthogs, similar to the domestic pig, are prone to produce pale,
soft and exudative (PSE) meat when exposed to ante mortem stress. As noted
by Somers (1997), in South Africa, as in other parts of Africa, poverty is
widespread and use of wildlife as a source of income and food is important to
local communities.
Hoffman et al (2005) have noted that warthog is a species regular consumed by
locals in South African restaurants and is also a game species frequently
consumed by tourists visiting South Africa (Hoffman et al 2003a). However,
no literature could be sourced where the nutritional composition of this species
has been reported. Therefore, the objective of the present investigation was to
collect base line data of the yield and chemical composition of the meat from
the warthog.
Materials and methods
Warthogs were hunted in July (they normally breed in the September to November,
with a peak in October in Southern Africa) in the Leeukop Nature Reserve (27˚25’
31˚55’) in KwaZulu Natal, South Africa. In this investigation, a single
sharp-shooter fired all the shots using a .243 Winchester fitted with a
telescopic sight and silencer. The shots invariably caused the remainder
of the herd (flounder) to run, although not at an alarming rate, still only one
pig was shot per herd. All the shots were fired in the daytime and
utilized a flank, neck or head-shot to kill the warthog. Directly after
shooting, the animals were exsanguinated by sticking.
Immediately after killing, the animals were weighed to obtain a hot body weight.
After the targeted number of hogs (n = 5) had been harvested the animals were
transported to a commercial small scale game processing plant where they were
further processed according to standard South African and Zimbabwean practices.
This consisted of skinning (it is standard practice to skin both warthogs and
wild pigs in Africa as very few game processing plants have the facility to
remove the hair), removal of the head, evisceration, and cleaning of carcasses
(Hoffman 2000). Most of the subcutaneous fat is removed with the skin (the
warthog is similar to the domestic pig in having a “thick” widely spread
subcutaneous fat depot). After cooling overnight
in a cooler (1 °C), the carcasses were cut into the following commercial cuts:
shoulder, loin, back, belly and hind legs. The hind legs were removed
after loosening the flanks on the inside of the legs (following the curve of the
leg muscles) to an imaginary line perpendicular to the ilium (seen from the
inside of the carcass). The leg was then removed by cutting along this
line, just missing the ilium (through the last lumbar vertebrae). The
weight of the two legs was recorded before the two legs were split along the
sacral vertebrae using a bandsaw. The rest of the carcass was halved prior
to separating into trade joints. The shoulder was removed by sawing along
an imaginary line from the elbow joint to a point below the spinal
column, between the 5th and 6th ribs. The
carcass was then swiveled so that the spinal column was sawn through at right
angles. The belly was removed by sawing from the Muscularis obliquus
abdominis internus
muscle parallel to the spine. The loin and back was separated
perpendicularly to the spinal column at the junction of the thoracic and lumbar
vertebrae. All commercial joints were weighed on a digital computing scale
(Digi DS-680) to the nearest gram.
The
pH and carcass temperatures were measured within 10 minutes of death, and 45
minutes thereafter at regular periods until the pH decline had reached a
plateau. The pH was measured in the lumbar region of the Musculus
longissimus dorsi using a calibrated (standard buffers at pH 4.0 and 7.0)
Crison 506 portable pH meter.
Thirty-six hours after cropping, two ca. 2 cm thick loin sub-samples (the first
at the first lumbar vertebrae and the second, caudally adjacent to it) were
taken for determination of drip and cooking loss (Honikel 1998). Drip loss
was expressed as the percentage moisture loss relative to the weight of the
fresh sample after a 48 hr hanging period in the cooler (1 ˚C). Cooking
loss (%) was determined by placing weighed samples of approximately 70 to 100 g,
sealed in plastic bags, in water at 70°C for 60 min. This allowed for sufficient
heat penetration without causing excessive denaturation of collagen present in
the meat. The bagged samples were allowed to cool down in running water to
ca. 25°C. Weight of the dried cooked samples were determined after
decanting the liquid phase, and the cooking loss calculated as total fluid lost,
expressed as a percentage of the fresh (uncooked) sample. Thereafter, five
1.27 cm diameter samples (from the centre of each Musculus longissimus
thoracis et lumborum
sample) were randomly removed for determining of Warner-Bratzler shear force
values. The samples were cut parallel to the muscle fibre direction in
order to measure the influence of the myofibrillar proteins. Maximum shear
force values (kg/1.27 cm diameter) were recorded for each sample (repeated five
times) and a mean was calculated for each individual animal. The colour of
the Musculus longissimus thoracis et lumborum was evaluated using a Color-guide
45º/0º colorimeter (Cat no: 6805; BYK-Gardner, USA) to determine L*, a* and b*
values (Commission International de l' Eclairage 1976) with L* indicating
brightness, a* the red-green range and b* the blue-yellow range.
The
rest of the loin sample (Musculus longissimus dorsi) was minced and used
for chemical analysis. Moisture content was determined by drying 2.5g
samples at 100 °C to constant weight, ashing at 500 °C for 5 h, protein (N x
6.25) content by the block digestion method (AOAC 1997). The total lipid
content was determined by chloroform/methanol solvent extraction (Lee et al
1996).
The
fatty acid and cholesterol content was determined as follows: After extracting
the lipids, fatty acid methyl esters (FAME) were prepared according to Morrison
and Smith (1964). The FAME were analysed with a GLC: Varian Model 3300,
equipped with flame ionization detection and two 30m fused silica megabore
DB-225 columns of 0.53 mm internal diameter (J & W Scientific, Folsom, CA).
Gas flow rates were: hydrogen 25 ml/min; air 250 ml/min and nitrogen (carrier
gas) 5-8 ml/min. The temperature program was linear at 4 ºC/min with
initial and final temperatures of 160 ºC and 220 ºC (held for 10 min),
respectively. The injector temperature was 240 ºC and the detector
temperature 250 ºC. The FAME were identified by comparison of the
retention times to those of a standard FAME mixture (Supleco TM 37 Component
FAME Mix, Catalogue Number 18919-1AMP, Lot number, LB-16064. Sigma Aldrich Inc.
North Harrison Road, Bellefonte, PA 16823-0048, USA).
Results and discussion
Carcass characteristics of
animals are presented in Table 1.
|
Table 1.
Mean
and standard deviation (SD) of weight (g) of carcasses, carcass
components, and by-products of warthogs (n=5) |
|
Measurement |
Weight,
g |
|
Mean |
SD |
|
Body weight |
61980 |
8548 |
|
Warm carcass |
32940 |
5047 |
|
Cold carcass |
32020 |
4949 |
|
Carcass
components |
|
|
|
Shoulder |
11970 |
2072 |
|
Loin |
2300 |
572 |
|
Back |
2760 |
390 |
|
Hind legs |
10270 |
1398 |
|
Belly |
4500 |
663 |
|
By-products |
|
|
|
Head |
7401 |
1055 |
|
Feet |
748 |
76.0 |
|
Skin |
3602 |
534 |
|
Liver |
817 |
134 |
|
Lungs |
836 |
273 |
|
Kidneys |
186 |
46.8 |
|
Heart |
221 |
31.5 |
|
Spleen |
59 |
9.5 |
|
Intestines |
12132 |
1917 |
|
Stomach |
1662 |
575 |
A dressing percentage, expressed as cold carcass weight as a
percentage of body weight, of 52 % was lower than 76 % obtained for Landrace X
Large White pigs that were reared in South Africa under either free-ranging
(body weight 86.4 kg) or conventional housing (body weight 78.7 kg) systems
(Hoffman et al 2003b). Furthermore, carcass weight in warthogs, in
contrast to domestic pigs, does not include the head, skin and adjacent
subcutaneous fat layers, accentuating the fact that dressing percentage is lower
in warthogs compared to domestic pigs. The head and skin form nearly 9 and
6 %, respectively, of the body weight of warthogs. Cold carcass weight
consists of 37 % shoulder and 14 % belly (Table 1), compared to 14 and 6 %,
respectively, for Landrace X Large White pigs (Hoffman et al 2003b) reared under
South African conditions. However, in contrast to the present study,
shoulder and belly percentages were determined without bone in the latter study.
A value of 15 % has been reported for loin as a percentage of cold carcass
weight in wild pigs shot in Poland (Mijewski and Korzeniowski 2000), compared to
7 % for warthogs found in the present study. Carcass weight in the former
study also included skin, as with domestic pigs. The above demonstrated
the difficulty in comparisons of warthog carcass characteristics with literature
values for other pig species due to differences in processing methodology.
Values for pH60min (Table 2) fall outside the
range considered critical (below 5.6) for developing PSE meat, and are also
below the critical limit (above 6.4) for dark, firm and dry (DFD) meat.
|
Table 2.
Mean
and standard deviation (SD) for physical characteristics of the loin
of warthogs (n=5) |
|
Characteristic |
Value |
|
Mean |
SD |
|
pH60min |
6.32 |
0.38 |
|
pH24h |
5.49 |
0.12 |
|
Drip loss
(%) |
2.76 |
1.32 |
|
Cooking loss
(%) |
16.79 |
3.39 |
|
Shear force
(kg/1.25 cm diameter) |
3.36 |
0.36 |
|
CIEL* |
46.37 |
2.53 |
|
CIEa* |
5.06 |
0.67 |
|
CIEb* |
9.21 |
0.76 |
Meat not subjected to either PSE or DFD has a pH24h
range of 5.5 to 5.8 (Lawrie 1985). However, an exponential decay model
fitted to the pH decline data (Wiklund et al. 1995) presented evidence that
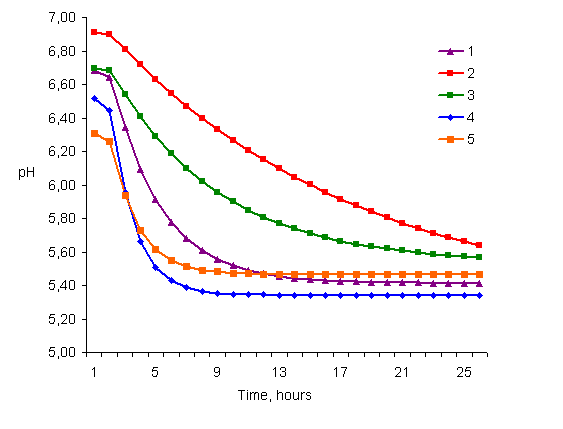
three of the five warthogs showed a rapid pH decline (Table 3; Figure 1), a
phenomenon associated with PSE type meat. Furthermore, there was a
positive correlation of 0.772 between the rate of pH change (c-value in the
exponential equation) and the percentage drip loss.
|
Table 3.
The effect of ante mortem stress on the rate of pH change and drip
loss of warthogs |
|
No |
Description of ante mortem stress |
Meat
Class |
Constants
for the exponential function (y = a + bect)* |
% Drip
loss |
|
a |
b |
c |
|
1 |
Shot behind
the shoulder, run 100m before dying |
Pale, soft,
exudative |
5.41 |
1.27 |
-0.31 |
3.35 |
|
2 |
Head shot,
died immediately |
Slightly
dark, firm, dry |
5.24 |
1.65 |
-0.06 |
2.45 |
|
3 |
Shoulder
shot, dropped immediately |
Normal |
5.53 |
1.17 |
-0.14 |
2.16 |
|
4 |
Head shot,
paralysed, frantic kicking movements |
Pale, soft,
exudative |
5.34 |
1.18 |
-0.65 |
6.76 |
|
5 |
Neck shot,
dropped immediately, frantic kicking movements |
Pale, soft,
exudative |
5.47 |
0.84 |
-0.58 |
3.14 |
|
*y
= pH at time t; a = ultimate pH; e = base of natural logarithm and b
and c are the function parameters describing the shape of the curve |
|
 |
|
Figure 1.
Post mortem pH change in the Musculus longissimus dorsi of
warthogs (warthogs 1, 4 and 5 showing a post-mortem pH decrease
characteristic to the pale, soft, exudative condition, warthog 2 a
pattern illustrating a slightly dark, firm, dry condition, and warthog 3
a pattern of normal meat) |
Values for warthog (Table 2) compare well to values of 6.16
and 5.53 reported for pH45min and pH24h, respectively, in
meat from Landrace X Large White pigs (Hoffman et al.2003b). Marchiori
and de Felício (2003) found values of 6.18 and 5.57 for pH60min
and pH24h, respectively, in the Musculus longissimus of wild
boars raised in Brazil (members of the European wild boar subspecies), Müller et
al (2000) determined a value of 5.45 for pH24h in wild pigs from
Germany, whereas Mijewski and Korzeniowski (2001) reported a value of 5.44 for
pH24h in meat of wild boars from Poland.
Under South African rearing conditions with similar
laboratory techniques for analysis, drip loss of 4.11 %, cooking loss of 22.59
%, CIEL* of 55.32, CIEa* of 2.81 and CIEb* of 10.10 have
been reported (Hoffman et al 2003b) in the Musculus longissimus lumborum
of the domestic pig. Similar drip losses (3.42 to 4.55 %), and higher
values for colour (CIEL* of 51.30, CIEa* of 7.94 and CIEb*
of 13.24) have been reported for Musculus longissimus of Brazilian wild
boars (Marchiori and de Felício 2003). Ranges for colour in normal meat
have been suggested as 52.2 to 54.8 for CIEL*, 5.1 to 7.5 for CIEa*,
and 12.9 to 14.5 for CIEb* (Van der Wal et al 1988). Although
it seems that meat from warthog has a lower lightness (CIEL*) than either
domestic or wild pigs, intermediary red colour intensity (CIEa*), and
yellow colour intensity similar to meat from domestic pigs (CIEb*),
direct comparisons are eliminated by differences in time post-mortem that
measurements have been taken.
Comparable to values reported in Table 4, values of 74.54,
1.82, 22.31 and 1.20 % for moisture, lipid, protein and ash, respectively, have
been determined in the Musculus longissimus lumborum of the domestic pig
in South Africa (Hoffman et al 2003b).
|
Table 4.
Mean
and standard deviation (SD) of chemical composition of the
loin of warthogs (n=5) |
|
Component |
% |
|
Mean |
SD |
|
Moisture |
74.04 |
0.94 |
|
Total lipid |
1.69 |
1.39 |
|
Protein |
22.14 |
0.30 |
|
Ash |
1.29 |
0.03 |
|
Fatty
acids |
|
|
|
C14:0 |
0.75 |
0.66 |
|
C16:0 |
19.95 |
2.25 |
|
C18:0 |
14.68 |
2.96 |
|
C20:0 |
0.14 |
0.02 |
|
C22:0 |
0.13 |
0.05 |
|
C24:0 |
0.10 |
0.09 |
|
Total SFA |
35.75 |
3.01 |
|
C16:1n7 |
0.74 |
0.76 |
|
C18:1n9 |
15.79 |
11.23 |
|
C20:1n9 |
0.07 |
0.06 |
|
C24:1n9 |
0.10 |
0.19 |
|
Total MUFA |
16.70 |
12.10 |
|
C18:2n6 |
26.12 |
9.64 |
|
C18:3n6 |
0.17 |
0.05 |
|
C18:3n3 |
7.26 |
7.97 |
|
C20:2n6 |
0.30 |
0.02 |
|
C20:3n6 |
1.06 |
0.60 |
|
C20:4n6 |
7.48 |
4.94 |
|
C20:3n3 |
0.94 |
0.35 |
|
C20:5n3 |
0.91 |
0.60 |
|
C22:2n6 |
0.07 |
0.16 |
|
C22:4n6 |
0.40 |
0.37 |
|
C22:3n3 |
0.00 |
0.00 |
|
C22:5n3 |
2.44 |
2.01 |
|
C22:6n3 |
0.42 |
0.35 |
|
Total PUFA |
47.56 |
10.35 |
|
SFA =
saturated fatty acids; MUFA = mono-unsaturated fatty acids; PUFA =
polyunsaturated fatty acids |
This is in agreement with values of 74.04, 1.95, 21.80 and
1.01 reported for wild pigs from Poland (Mijewski and Korzeniowski 2001),
although it was not mentioned if fat extraction in the latter study was
performed by ether or methanol/chloroform extraction.
The most abundant fatty acid found in warthog meat was the
polyunsaturated linoleic (C18:2n-6) acid (Table 4). Not only the amount of
fat consumed, but also its saturation is important in prevention of coronary
diseases (Girolami et al 2003). Fatty acid composition of dietary fat
influence cholesterol levels in humans in that high dietary level of long-chain
saturated fatty acids increase plasma cholesterol levels compared with high
levels of mono- and polyunsaturated fatty acids (Grundy and Denke 1990).
With a similar fatty acid analysis than technique to that used in the present
study (Table 4), Hoffman et al (2003b) found contents of
saturated, mono-unsaturated, and polyunsaturated fatty acids as 42.39, 39.19
and 18.43 %, respectively, in the Musculus longissimus lumborum of
Landrace X Large White pigs kept under conventional housing conditions in South
Africa. A similar tendency has been found when domestic pigs have been
kept under free-ranging conditions. This clearly illustrates the influence
of diet on meat fatty acid composition, which is especially profound in
monogastric animals (Wood et al 2004). The polyunsaturated: saturated
fatty acid ratio in meat from warthogs was found to be 1.33 (Table 4), compared
to 0.46 to 0.64 in domestic pigs (Hoffman et al 2003b). Whereas Raes et al
(2004) suggested a polyunsaturated: saturated fatty acid ratio of 0.7 or higher
in meat, Wood and Enser (1997) recommended a ratio of 0.45 as the minimum.
Conclusions
-
It can be concluded from this study that warthogs can be used
as a meat source for human consumption.
-
Meat from this species is not only
comparable that that of other pig species regarding physical meat
characteristics, but also supply a meat that can be promoted on a favourable
fatty acid profile for human health concerns.
-
Presently, warthogs are
found in abundance within southern Africa and are readily consumed within the
local restaurant trade. There are also no hunting restrictions on this species.
-
The only restriction on this species according to the Animal Diseases Act (Act
35 of 1984) and its regulations is the movement of carcasses out of African
Swine Fever control areas.
References
AOAC
1997
Official method of analysis. 16th
edition. Association of Official Analytical Chemists, Inc. Arlington,
Virginia, USA
Commission Internationale de I’Eclairage 1976
Colorimetry, 2nd edition.
Vienna, Switzerland: CIE
Girolami A, Marsico I, D’Andrea G, Braghieri A, Napolitano F and Cifuni G F 2003
Fatty acid profile, cholesterol content and tenderness of ostrich
meat as influenced by age at slaughter and muscle type. Meat Science 64: 309-315
Grundy
S M and Denke M A 1990
Dietary influences on serum lipids. Journal of Lipid Research 31:
1149–1172
Hoffman L C 2000
The yield and carcass chemical composition of impala (Aepyceros melampus),
a southern African antelope species. Journal of the Science of Food and
Agriculture 80: 752-756
Hoffman L
C 2001
The effect of day-time cropping on warthog (Phacochoerus
africanus) meat quality. 47th International Congress of Meat Science and
Technology. Krakow, Poland (26-31 August). Volume
II. p. 70-71
Hoffman L C, Crafford K, Muller N and Schutte D-W 2003a
Perceptions and consumption of game meat by a group of tourists visiting South
Africa. South African Journal of Wildlife Research 33: 125-130
Hoffman L
C, Styger E, Muller M, Brand T S
2003b
The growth and carcass and meat characteristics of pigs raised in a free-range
or conventional housing system. South African Journal of Animal Science 33:
166-175
Hoffman L C, Muller M, Schutte D W and Crafford K 2005
Consumer expectations, perceptions and purchasing of South African game meat.
South African Journal of Wildlife Research 35: 33-42
Honikel K O 1998
Reference methods for the assessment of the physical characteristics of meat.
Meat Science 49: 447-457
Lawrie R A 1985
Meat Science, 4th edition. Pergamon
Press, Oxford, United Kingdom
Lee C
M, Trevino B and Chaiyawat M 1996
A simple and rapid solvent extraction method for determining
total lipids in fish tissue. Journal of AOAC International 79: 487-492
Marchiori A F and de Felício P E 2003
Quality of wild boar meat and commercial pork. Scientia Agricola 60: 1-5
http://www.scielo.br/pdf/sa/v60n1/14535.pdf
Mason D R 1985
Postnatal growth and physical condition of warthogs Phacochoerus africanus
in Zululand. South African Journal of Wildlife Research 15: 89-97
Mijewski T and Korzeniowski W 2000
Tissue composition of wild boars carcasses. Electronic Journal of Polish
Agricultural Universities, Food Science and Technology, Volume 3, Issue 2.
Available Online
http://www.ejpau.media.pl/series/volume3/issue2/food/art-03.html
Mijewski T and Korzeniowski W 2001
Technological properties of wild boars meat. Electronic Journal of Polish
Agricultural Universities, Food Science and Technology, Volume 4, Issue 2.
Available Online
http://www.ejpau.media.pl/series/volume4/issue2/food/art-02.html
Morrisson W R and Smith M L 1964
Preparation of fatty acid methyl esters and dimethylacetals from lipids with
boron fluoride-methanol. Journal of Lipid Research 5:600-608
Müller E, Moser G, Bartenschlager H and Geldermann H 2000
Trait values of growth, carcass and meat quality in Wild Boar, Meishan and
Pietrain pigs as well as their crossbred generations. Journal of Animal Breeding
and Genetics 117:189–202
Raes K, Haak L, Balcaen A, Claeys E, Demeyer D and De Smet S 2004
Effect of linseed feeding at similar linoleic acid levels on the fatty acid
composition of double-muscled Belgian Blue young bull. Meat Science 66: 307-315
Skinner J D and Smithers R H N 1990
The mammals of the southern African subregion. University of Pretoria, Pretoria,
South Africa
Somers M J and Penzhorn B L 1992
Reproduction in a reintroduced warthog population in the eastern Cape Province.
South African Journal of Wildlife Research 22: 57-60
Somers M J 1997
The sustainability of harvesting a warthog population: assessment of management
options using simulation modelling. South African Journal of Wildlife Research
27: 37-43
Van der Wal P G, Blink A H and Merkus G S M 1988
Differences in quality characteristics of normal, PSE and DFD
pork. Meat Science 24: 79-84
Wiklund E, Andersson A, Malmfors G, Lundström K and Danell Ö 1995
Ultimate pH values in reindeer meat with particular regard to animal sex and
age, muscle and transport distance. Rangifer 15: 47-54
Wood J D and Enser M 1997
Factors influencing fatty acids in meat and the role of antioxidants in
improving meat quality. British Journal of Nutrition 78: S49-S60
Wood J D, Richardson R I, Nute G R, Fisher A V, Campo M M,
Kasapidou E, Sheard P R and Enser M 2004
Effects of fatty acids on
meat quality: a review. Meat Science 66: 21-32
Received 11 February 2007; Accepted 14 March 2007; Published 5 October 2007
Go to top
